Millions of Americans suffer from respiratory diseases, including Asthma and chronic obstructive pulmonary disease (COPD). Together, these two diseases affect the day-to-day lives of over 40 million patients in the U.S. Furthermore, respiratory allergies affect 10 percent of children under the age of 18. Not only do these diseases impact day-to-day functioning, but they can also be life-threatening.
Today, America’s biopharmaceutical companies are developing 130 medicines for the millions of patients affected by respiratory diseases. All these medicines in development are currently awaiting review by the U.S. Food and Drug Administration (FDA) or in clinical trials:
- 54 medicines for asthma: The most common chronic condition among children in the U.S., with more than 6 million children and 20 million adults affected.
- 39 medicines for COPD: Affecting approximately 16 million Americans, these medicines also include treatments for emphysema and chronic bronchitis.
12 medicines for allergies with respiratory symptoms: The sixth leading cause of chronic illness in the U.S., common allergens can range from peanuts, to dust mites and tree pollen.
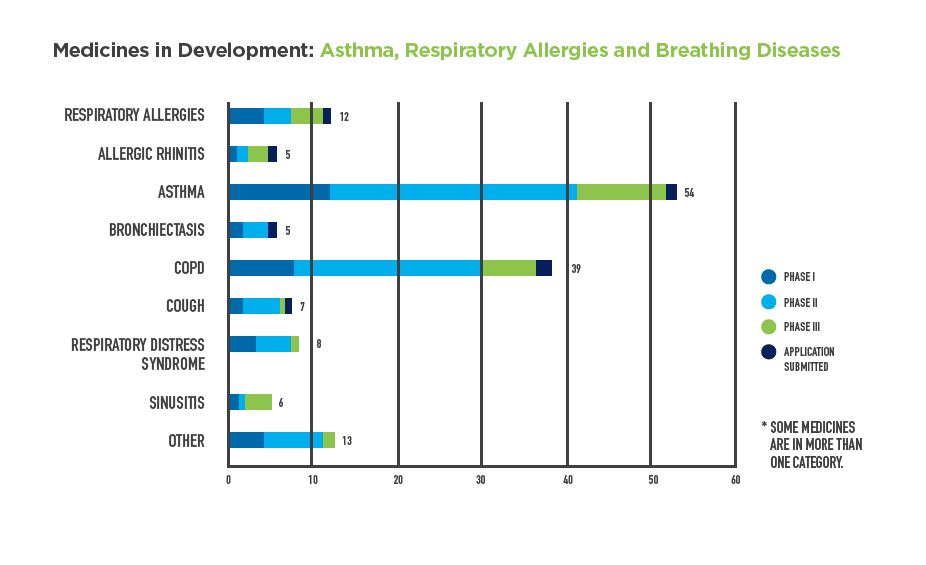 These treatments represent significant advances in the biopharmaceutical pipeline. Notably, among the 130 medicines in development is a potential first-in-class treatment for asthma, a COPD medicine targeting lung inflammation and a combination therapy treatment for peanut allergies.
These treatments represent significant advances in the biopharmaceutical pipeline. Notably, among the 130 medicines in development is a potential first-in-class treatment for asthma, a COPD medicine targeting lung inflammation and a combination therapy treatment for peanut allergies.
Recent advancements in science and technology have altered the way we define disease, develop medicines and prescribe treatments. In particular, personalized medicine is helping patients receive effective treatment by leveraging the individual’s unique information to better diagnosis and treat for that patient. In both asthma and COPD, the role of genetics and phenotypes offer to play an important role in diagnosing and treating respiratory diseases through personalize medicine.
The 130 medicines in development by America’s biopharmaceutical companies represent critical advances in our understanding of respiratory illnesses and in improving quality of life for millions of Americans. To learn more about these potential new treatments, view the full report here.



 These treatments represent significant advances in the biopharmaceutical pipeline. Notably, among the 130 medicines in development is a potential first-in-class treatment for asthma, a COPD medicine targeting lung inflammation and a combination therapy treatment for peanut allergies.
These treatments represent significant advances in the biopharmaceutical pipeline. Notably, among the 130 medicines in development is a potential first-in-class treatment for asthma, a COPD medicine targeting lung inflammation and a combination therapy treatment for peanut allergies.