5 Reasons why biopharmaceutical patents are different
Intellectual property protection has deep, historical roots in the United States.
5 Reasons why biopharmaceutical patents are different.
Intellectual property protection has deep, historical roots in the United States.

5 Reasons why biopharmaceutical patents are different.
Intellectual property protection has deep, historical roots in the United States. Article I, Section 8 of the U.S. Constitution expressly recognizes the role of patents to “promote the Progress of Science and useful Arts.” It was under this Constitutional authority that the Congress enacted the basic Patent Act of 1793, authored by Thomas Jefferson. A half-century later, Abraham Lincoln said patents add “the fuel of interest to the fire of genius.”
Since the founding of our country, legal protection for innovation and creativity has been the key to critical advances in science, technology and arts that have generated economic growth, benefited society and improved the quality of life for all Americans.
In medicine, it is the patent system that fulfills the promise of breakthroughs in treatments and cures for AIDS, tuberculosis and scores of debilitating or life-threatening illnesses around the world.
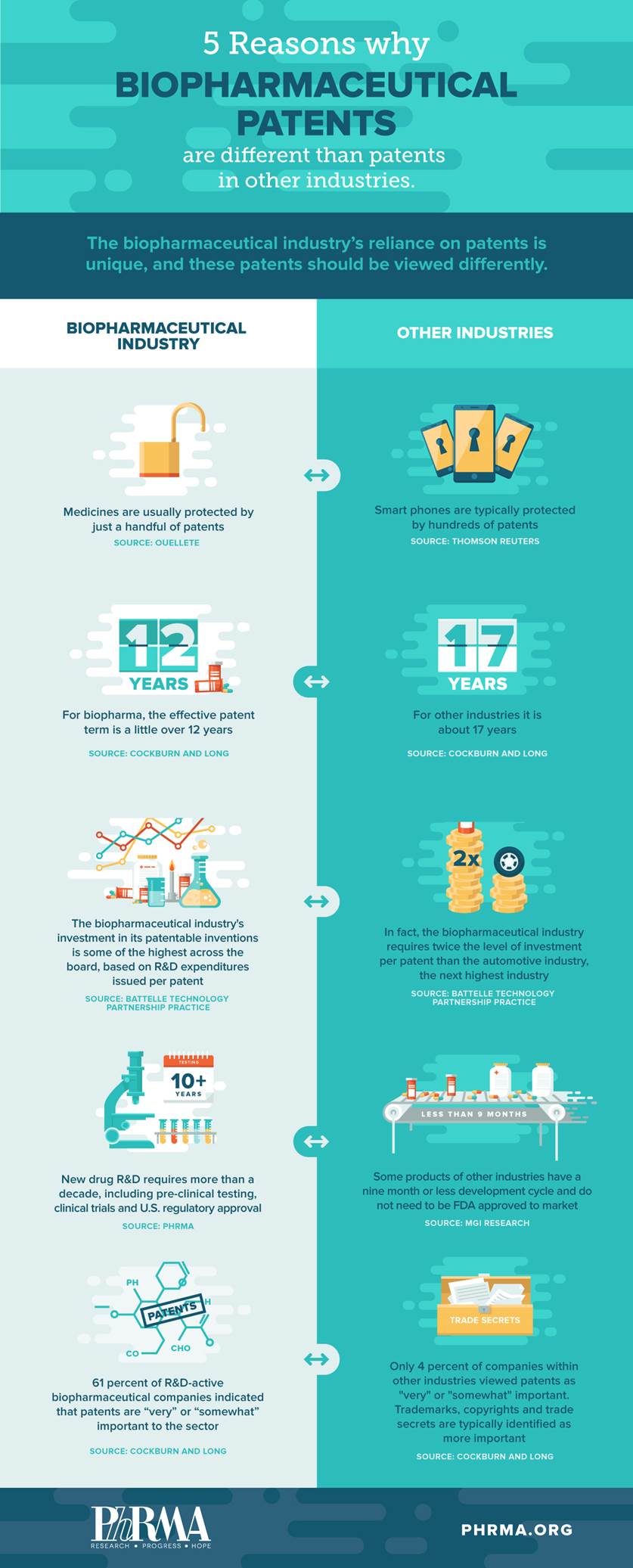
Patents are the “lifeblood” of the pharmaceutical industry. And they are not like the patents in other industries. Here are five reasons how:
To learn more about how biopharmaceutical patents are different, check out our new infographic here: