A Conversation on Continuing to Improve the Lives of Patients Living with Arthritis
For many patients, hope for a brighter future is directly tied to the potential of medicines in the pipeline to treat their disease or condition. The light at the end of the tunnel is a little brighter today with 92 medicines in development by biopharmaceutical research companies for the more than 52 million Americans with some form of arthritis or musculoskeletal disease. These medicines, according to a new Medicines in Development report by PhRMA, are either in clinical trials or awaiting review by the U.S. Food and Drug Administration (FDA) and offer great promise.
Each year, arthritis and related conditions account for 44 million outpatient visits and nearly one million hospitalizations. The 92 medicines in development target gout, juvenile arthritis, musculoskeletal pain, osteoarthritis, psoriatic arthritis, rheumatoid arthritis (RA), and spondylitis. Right now, 155 clinical trials are taking place to study new treatments targeting arthritis, and 97 of them have just begun to enroll patient participants. The development of new, innovative therapies for arthritis or any other disease would not be possible without the patients who volunteer to participate in clinical trials.
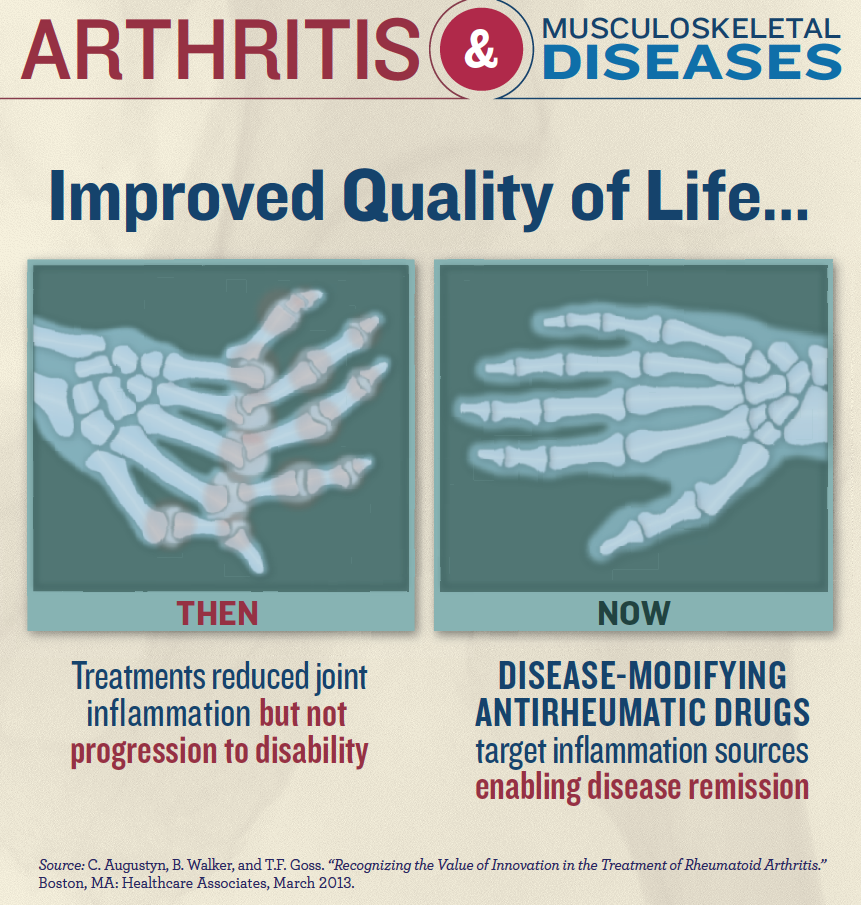
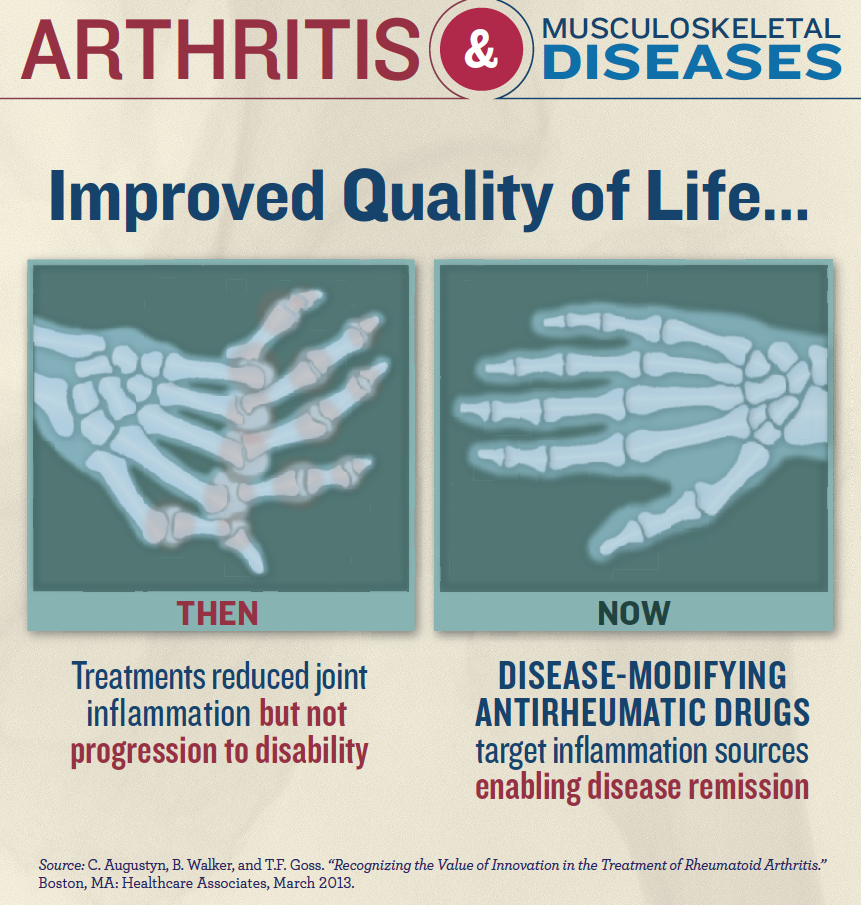
Important progress has already been made toward treating arthritis. For example, combination therapy has provided patients with RA a 35 percent chance of complete clinical remission over the course of five years, according to a 2010 study in the Journal of Rheumatology. A mere 20 years ago, the disease was viewed as relentlessly progressive and came with a bleak prognosis. Now, many patients are living their lives symptom-free.
Innovations like those for RA patients drive biopharmaceutical companies to keep searching for the next major breakthrough. We posed the following question in our Conversations forum:
“We’ve seen great strides to treat arthritis and related diseases. How do we continue to make headway to improve the lives of patients?”
The medicines in the pipeline provide hope that successful treatment options will be available to those living with these conditions. We look forward to hearing from the contributors and from you about how we can continue the progress in helping patients with arthritis and related diseases.
Please share your thoughts to this important dialogue online below or by joining the conversation on Facebook or Twitter.