More than 6 million Americans, most of them age 65 or older, live with Alzheimer’s disease, a devastating degenerative brain disease and the most common form of dementia. After heart disease and cancer, Alzheimer’s disease is the third leading cause of death for seniors, responsible for one out of every three deaths. Not only do patients need an effective treatment and cure, but the health care system does, too. Alzheimer’s burden is expected to cost the nation more than $1.1 trillion by 2050.
Currently, there are five Food and Drug Administration (FDA) approved medicines to treat the disease, but these medicines only partially manage symptoms by helping to maintain mental function and control behavioral challenges. There remains a substantial unmet medical need for disease-modifying treatments that can prevent, slow or cure the disease.
Research into neurological diseases like Alzheimer’s has been exceptionally challenging and characterized by many setbacks. Several unique challenges in this space include:
- Limitations of preclinical models which can hamper the entry of drug targets into Phase 1 clinical testing.
- Absence of validated, non-invasive biomarkers for early diagnosis, a challenge because of the complexity in identifying the disease.
- Clinical trial challenges that can range from participation to adequate monitoring.
- A significant gap in scientific knowledge due to the complexity of the brain.
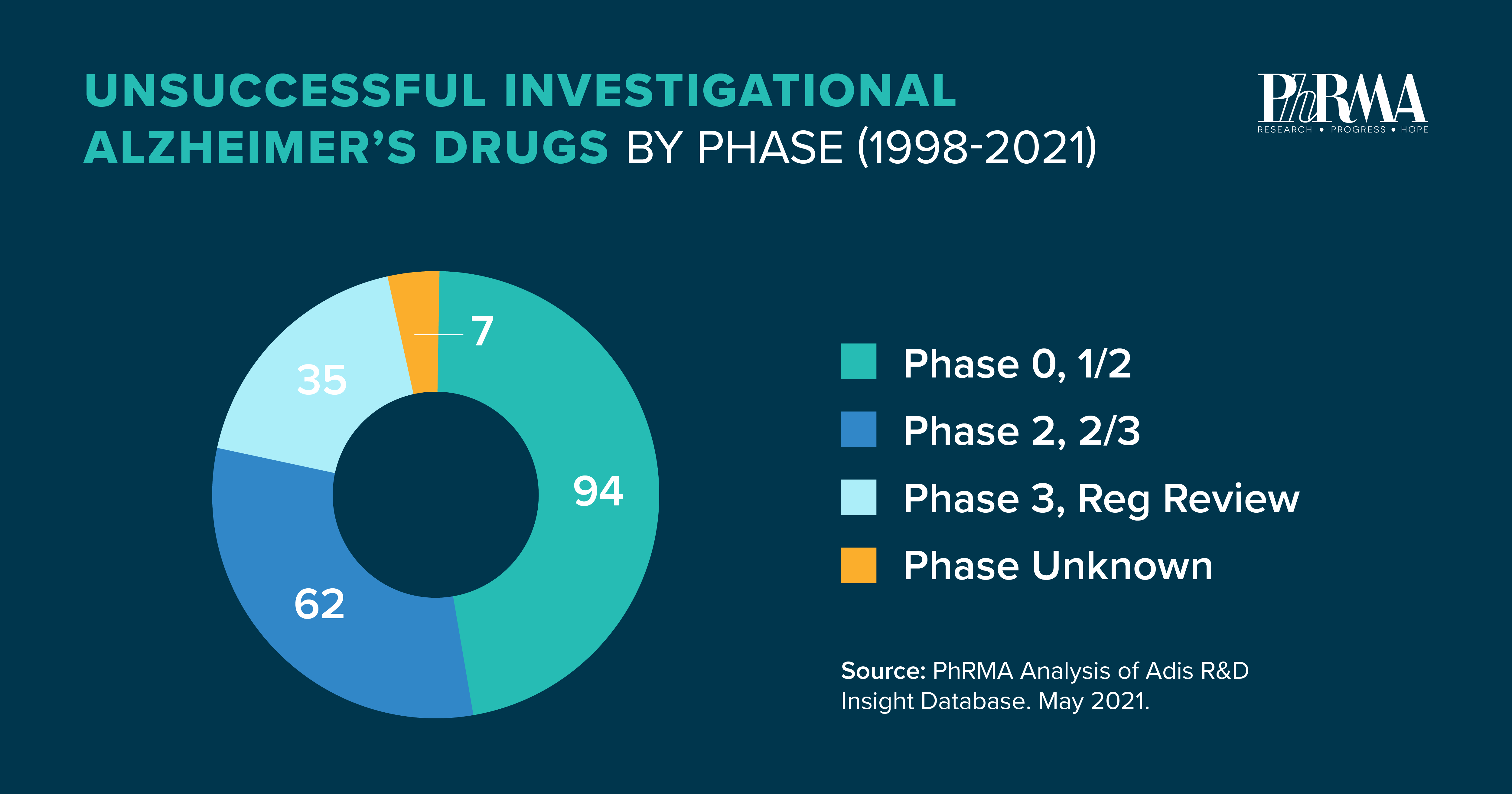
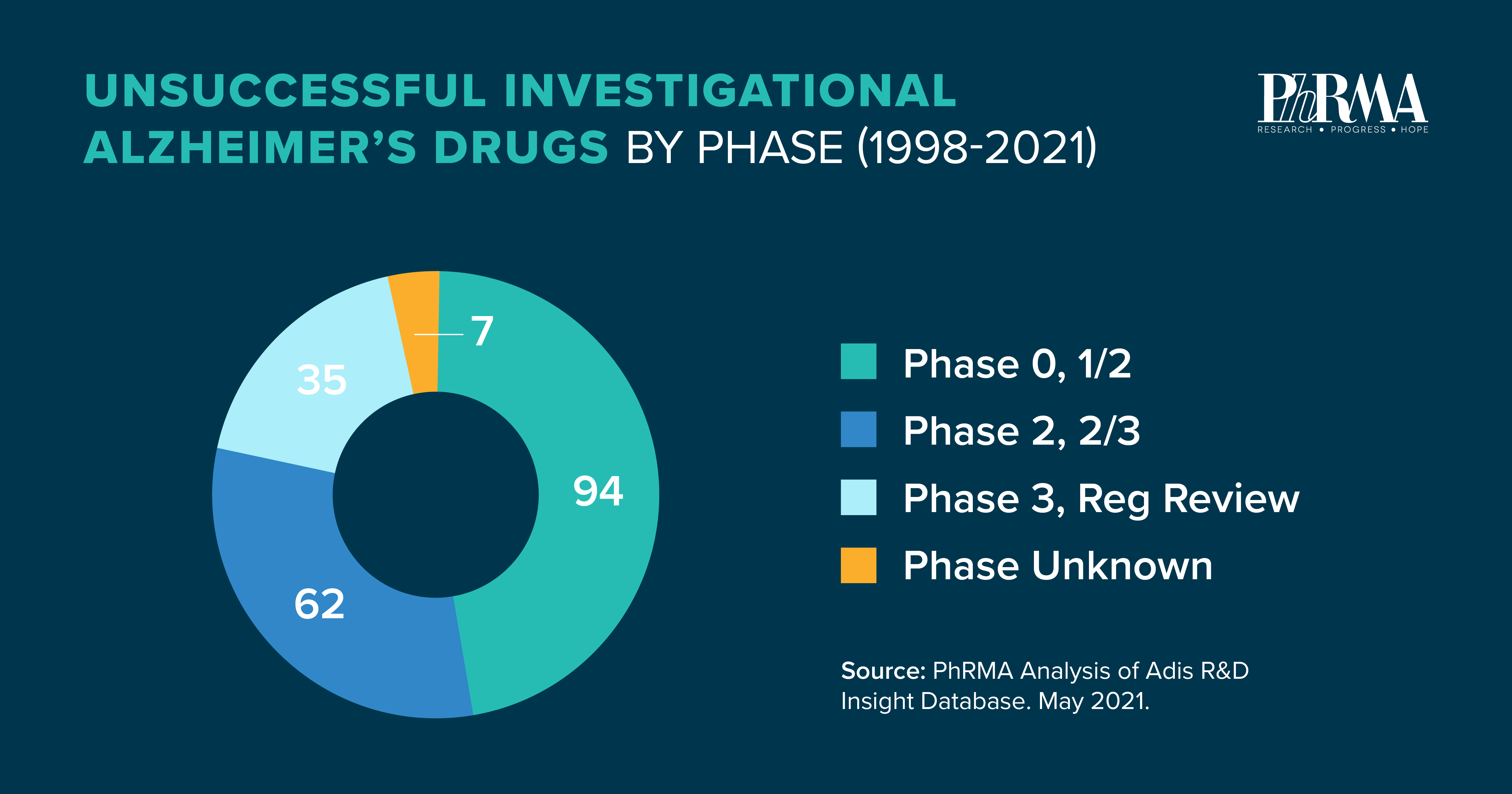
Despite the immense challenges, biopharmaceutical companies remain committed. Medical research is most often iterative, with breakthroughs rarely happening overnight and lessons learned from unsuccessful projects helping to inform new avenues of research while also providing critical information to other potential treatments under investigation.
Building from lessons learned from efforts to date, biopharmaceutical researchers are currently researching more than 80 potential medicines to attack the disease in diverse, innovative ways that researchers hope will halt or prevent Alzheimer’s. Approximately 82.5% have the potential to be disease modifying treatments.
Due to recent advances in understanding the nature of the disease and learning from previous clinical research, there is immense, growing hope for treatments and cures that can curb the impact of this disease. One study found that, by 2050, the number of people living with Alzheimer’s disease could be reduced by 40% if a new treatment that is able to delay disease onset by five years is approved by 2025. The same study found a treatment of this kind could yield an estimated $367 billion in health care savings annually — including $141 billion in savings to Medicare and $77 billion to Medicaid. Other cost savings and relief could come to the over 11 million caregivers across the United States, who are disproportionately women. Unpaid caregivers provided an estimated 15.3 billion hours of care valued at nearly $257 billion last year.
While biopharmaceutical companies continue to research treatments for Alzheimer’s, the industry’s ability to develop complex therapies for diseases like Alzheimer’s in the future are being threatened by harmful pricing policy proposals. Instead, policymakers should take a holistic approach to lowering patient costs while supporting medical innovation and ensuring Americans have access to the best treatments available. This environment is critical to support the continued innovation that patients with Alzheimer’s and other diseases so desperately need.
To learn more read the full fact sheet here.