The United States biopharmaceutical industry continues to innovate and discover critical solutions for our toughest health challenges such as the COVID-19 pandemic. America’s global leadership has not been by chance, but rather the result of a well-planned effort to facilitate innovation by incentivizing and protecting inventors and supporting collaboration between and among the public and private sectors.
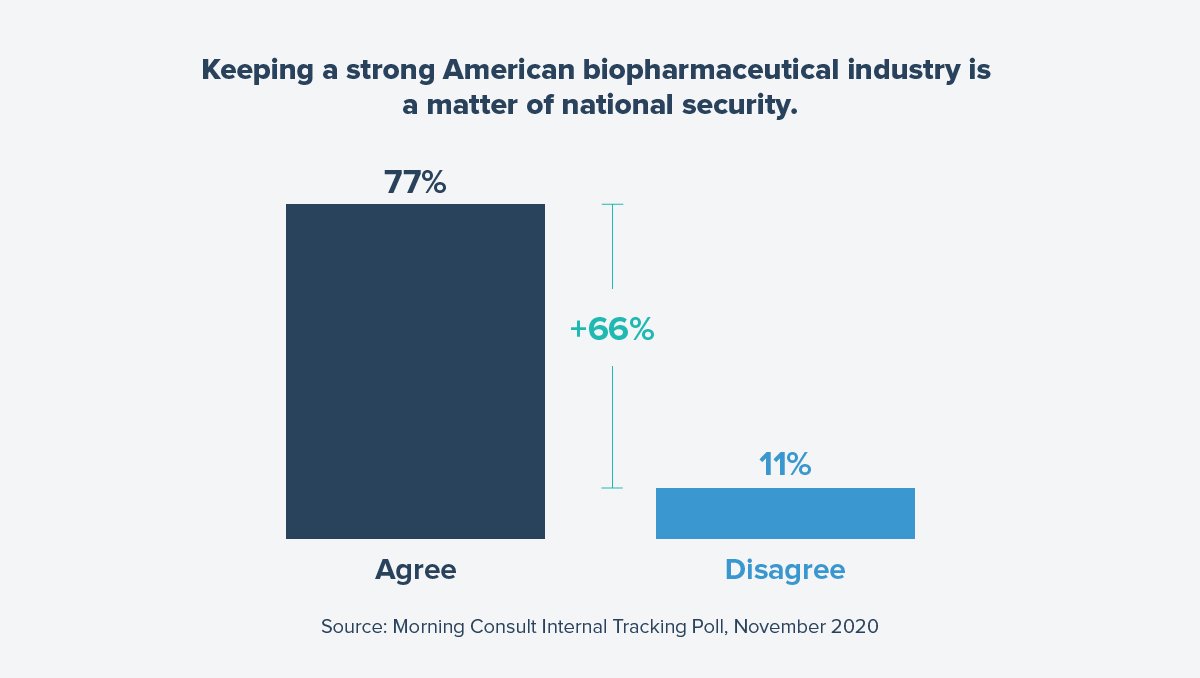
Americans appreciate industry’s role in advancing innovation. In fact, 62 percent of Americans viewed the industry positively in February – a number 30 points higher than in January 2020. Further, 77% agree that keeping a strong American biopharmaceutical industry is a matter of national security to protect us against the next pandemic.
But how did we get here? Consider current public sentiment in the U.S. and how it ties back to policies and investments put in place – in some cases – more than 40 years ago.
87% of Americans agree the coronavirus pandemic has shown how important it is for drug companies to make significant investments in research and development, according to a Public Opinion Strategies internal poll late last year.
- This biopharmaceutical industry leadership is, in part, tied to the Bayh-Dole Act of 1980, as well as other legal and regulatory frameworks. Bayh-Dole specifically allows federally funded researchers to patent inventions and license those patents to companies who then invest in further research and development – leveraging capabilities in an effort to commercialize a product for patients. Such protections and policy frameworks have led to an innovation ecosystem that generations more new medicines than any other country in the world – for example 53 novel drug approvals in 2020 including the first approved treatment for COVID-19.
- Importantly, this innovation “engine” has an exponential ability to help spur future breakthroughs. Consider the COVID-19 vaccines using mRNA and viral vector technology; they mark a new beginning for vaccines and combatting viruses and other illnesses, such as cancers, and open an entirely new world of insights and discoveries.
The Harris Poll noted fear about the vaccine rollout being too slow dropped from 47% in January to 36% in March. Among the likely contributing factors is the biopharmaceutical’s ability to leverage advanced manufacturing capabilities that have accelerated production and distribution.
- A strong innovation ecosystem enables collaboration on everything from discovery of new medicines to distribution of those medicines across the country and world. Thus, the country is well poised to continue to innovate and drive needed solutions, such as more efficient vaccine delivery. In addition, investments in advanced biopharmaceutical manufacturing and infrastructure facilitate innovation delivery from the lab to the patient. Long recognizing that biopharmaceutical manufacturing provides the critical link between the discovery of a medicine and its availability to patients, the U.S. biopharmaceutical industry has heavily invested in manufacturing capabilities to pave the way for this innovation; the industry’s capital investments in facilities increased by 17% from 2010 to 2016. The resulting capabilities differentiates the country among most other markets worldwide, providing not only resources to meet patient needs but skilled jobs and a strong economic driver.
The U.S. system of developing medicine is unique – it relies on a mix of private sector, public sector and research institutions working together. While not perfect, a dedicated set of actions to invest in innovation has helped the U.S. biopharmaceutical industry become a true strategic asset domestically – one that drives innovation with the ability to effectively address major crises, as with COVID-19. Data shows that Americans want a strong biopharmaceutical industry – supporting policies to facilitate a strong innovation ecosystem will further U.S. leadership and lead to a positive impact on patients worldwide.




