
The extraordinary financial pressure COVID-19 is creating for the U.S. economy underscores the need for a renewed push toward a more value-driven health care system that prioritizes patient access and affordability. Now more than ever, we need to make sure we’re spending our health resources wisely. That’s why it’s heartening to see the number of innovative contracts grow between biopharmaceutical companies and insurance companies as potential solutions for better affordability and better outcomes.
From 2009 through the first half of 2020, the number of publicly announced innovative contracts grew from two to 77. These contracts were undertaken by over two dozen biopharmaceutical companies and cover 49 brand medicines that address a wide range of diseases, including treatments for debilitating chronic conditions like multiple sclerosis and rheumatoid arthritis, as well as cutting-edge gene therapies for rare genetic disorders.
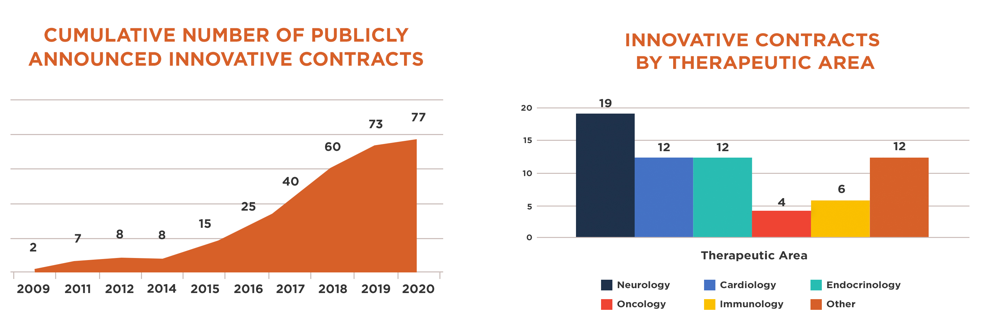
Innovative contracts help reduce cost pressures on the health care system by basing payment for medicines on how well they work for patients – as is the case with value-based contracts – or by allowing for a more flexible and sustainable approach to payment – such as with alternative financing arrangements like pay-over-time or subscription models. And by alleviating cost challenges for the system, they may enable broader patient access to life-saving medicines. In some cases, they’ve been shown to lower out-of-pocket costs for patients, which is particularly important given recent data indicate an upward trend in patient cost-sharing for brand medicines.
On top of the potential cost-saving benefits for patients and the system, innovative contracts can enable patient access without disrupting incentives for future innovation, which is essential as biopharmaceutical companies work around the clock to develop new treatments and vaccines to beat COVID-19.
As we seek to move our health care system from a fee-for-service model towards a more resilient, value-driven health care system that is better prepared for the next health crisis, it’s important that we pursue and advance policy solutions that improve access while maintaining incentives for the biopharmaceutical innovation.
To learn more, visit www.phrma.org/value-collaborative.





