Did you know that if you look at medicines in the pipeline for previously unmet medical needs, many are new and exciting immunotherapy treatments that are physician administered? That means these innovative medicines will fall under Part B coverage after Food and Drug Administration approval. Between 2006 and 2013, for example, there were 83 new medicines approved that are now covered by Part B, many of which are used to treat cancer or blood diseases. And a report out last year found there are more than 240 new immuno-oncology treatments in development.
In covering these types of physician-administered medicines, the Part B program supports some of the sickest and most vulnerable patients. These patients often suffer from diseases like cancer, rheumatoid arthritis, and macular degeneration – conditions that may be more common or complex to treat among older Americans enrolled in Medicare – and they rely on Part B coverage to access the needed treatments. For these patients, the promise of new, innovative medicines often provides much-needed hope.
This, though, has prompted policymakers to raise concerns about cost growth in the Part B benefit. However, year after year, as new Part B medicines have become available to beneficiaries, drugs have remained a small and stable share of overall spending.
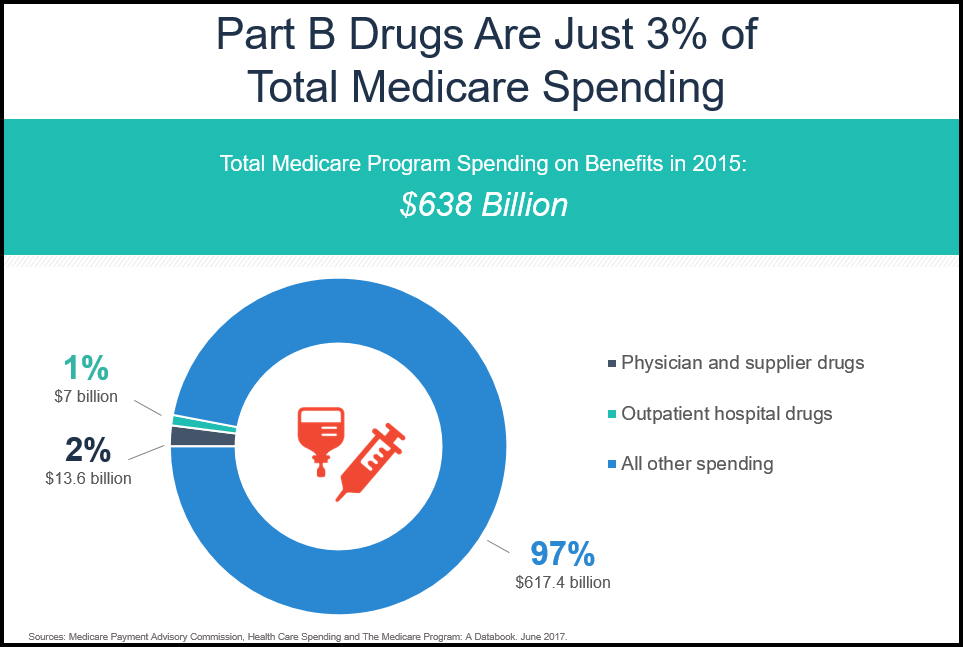
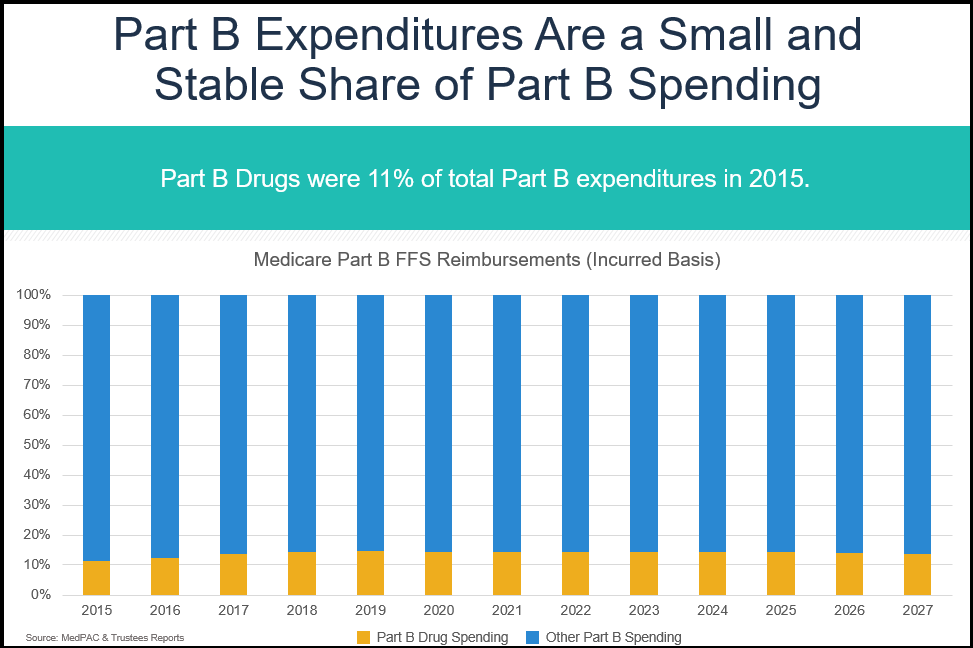
This was confirmed this past summer when the Medicare Payment Advisory Commission (MedPAC) released its annual report looking at health care spending relating to Medicare. The report found that in 2015, Part B drug spending grew modestly, but remained just 3 percent of total Medicare spending and 11 percent of spending under the Part B benefit.
This is consistent with MedPAC’s June 2016 Data Book that looked at 2014 data and also found that Part B drug spending was just 3 percent of total Medicare spending. Moving forward, experts project that drugs will continue to be a small and stable share of the Part B benefit. It is also important to note that the Part B program continues to evolve as it is incorporated into innovative payment designs like Accountable Care Organizations and the Oncology Care Model.
As conversations around drug spending and Part B continue, it is important to get the facts straight and to put them in the right context. Patients are counting on it.
Learn more at PhRMA.org/PartB.





