ICER framework threatens to halt continued progress against multiple myeloma
The ICER value framework threatens to halt continued progress against multiple myeloma.

ICER framework threatens to halt continued progress against multiple myeloma.
The ICER value framework threatens to halt continued progress against multiple myeloma.

ICER framework threatens to halt continued progress against multiple myeloma.

Progress in the fight against multiple myeloma, a cancer of the blood and bone marrow, has been nothing short of remarkable.
Just a decade ago, patients diagnosed faced a poor prognosis and treatment options were largely limited to chemotherapy, often associated with severe side effects.
Now, thanks in large part to biopharmaceutical discovery and innovation, multiple myeloma is a very treatable disease for many patients and survival rates for the approximately 27,000 Americans diagnosed each year have improved dramatically. Today, more than 80 percent of newly diagnosed patients are living more than four years. In 2015 alone, four new medicines were approved to treat this rare form of blood cancer, including two first-in-class therapies, offering patients entirely new ways to treat their disease.
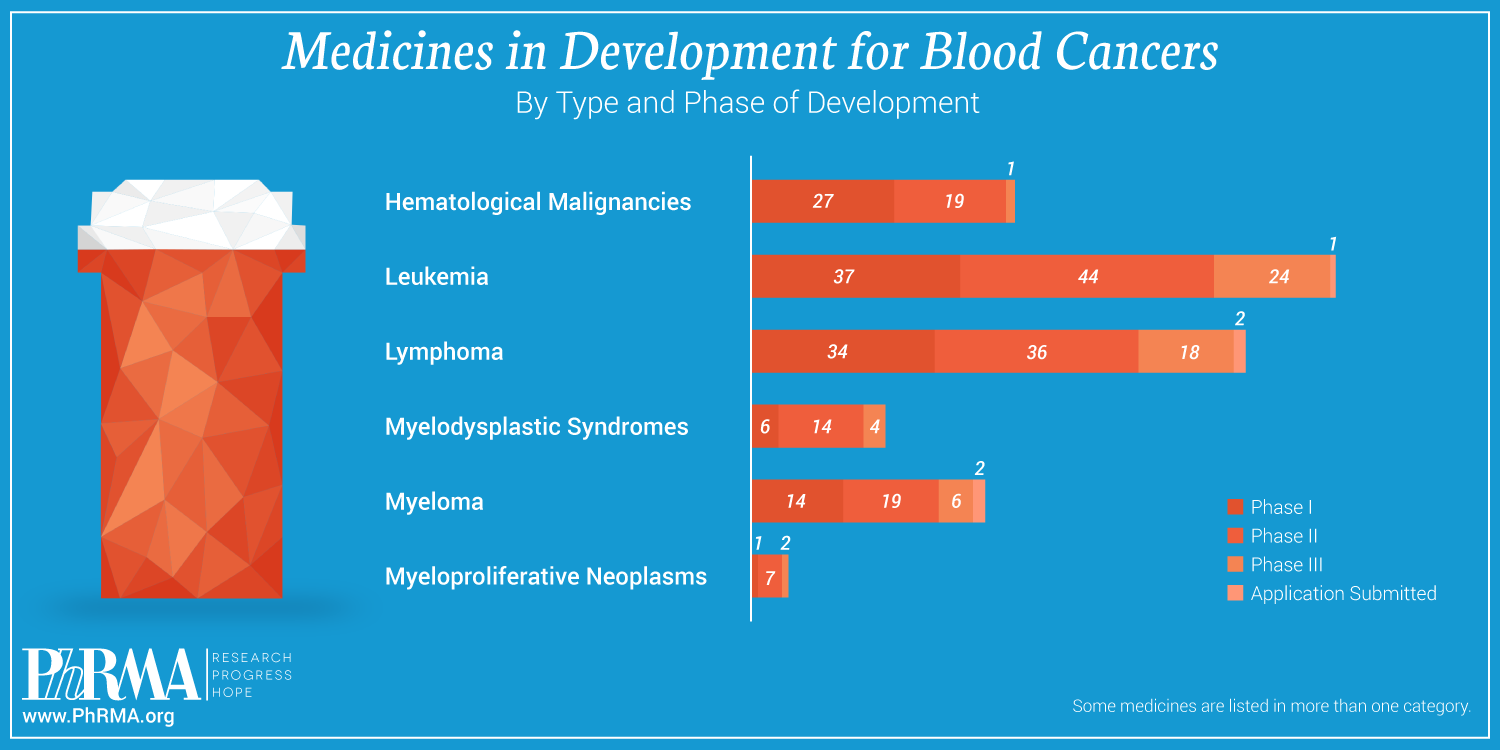
Biopharmaceutical researchers hope that the 41 medicines in development for multiple myeloma could help increase the survival rate. We look forward to hearing more at next week’s annual meeting of the American Society for Clinical Oncology about continued advances in treating multiple myeloma.
In contrast, an advisory panel made up largely of payers will assess multiple myeloma treatments using a flawed value framework developed by the Institute for Clinical and Economic Review (ICER) that threatens to put the brakes on continued progress against multiple myeloma.
When well-structured and used appropriately, frameworks to assess the value of medical tests, treatments and health care services can be useful in the private market to support patient-centered health care. That is why we are proud to support work by organizations such as the International Society for Pharmacoeconomics and Outcomes Research and the National Health Council to advance better approaches to value assessment.
However, the model developed by ICER uses methodology that is systematically biased against continued progress, devaluing advances through blunt budget caps and one-size-fits-all value thresholds that ignore the complex realities of optimally caring for cancer patients in an era of personalized medicine.
As highlighted by the make-up of ICER’s St. Louis advisory panel, the ICER model chooses payer needs over patients’ needs. Like the advisory panel, ICER’s Governing Board also is dominated by payer representatives.
While public stakeholders were given only two weeks to provide input on ICER’s 172-page report, that hasn’t stopped them from weighing in. Here is what patient and provider groups are saying about ICER’s assessment of multiple myeloma treatments:
ICER’s framework does not account for the aspects of value that matter to patients.
ICER’s framework does not accurately reflect how physicians treat patients and could be used to dictate physician prescribing and patient access.
ICER’s framework, which relies on a QALY-based cost-effectiveness analysis, conflicts with the movement toward personalized medicine.
Learn more about how ICER’s model doesn’t meet patients’ needs and support continued improvement in health care here.