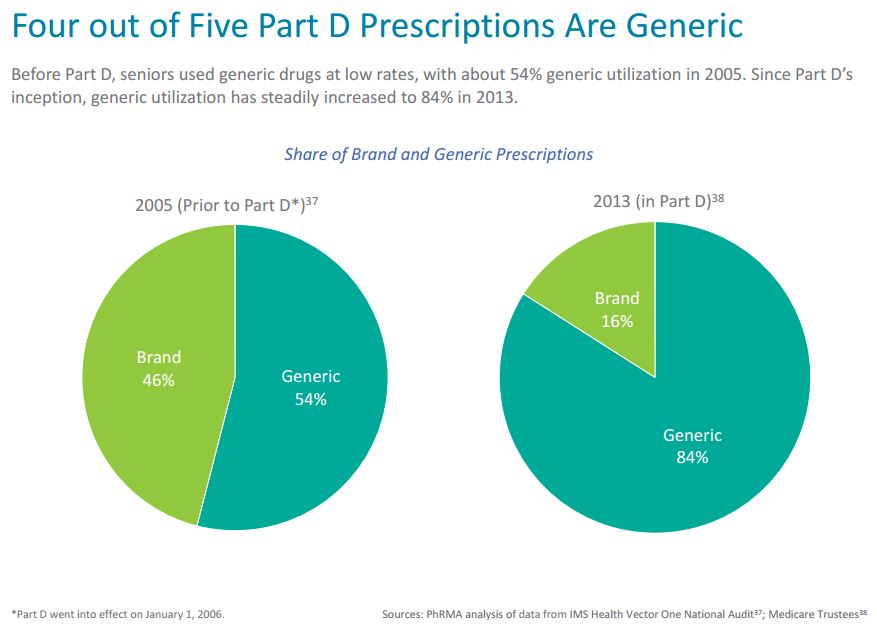
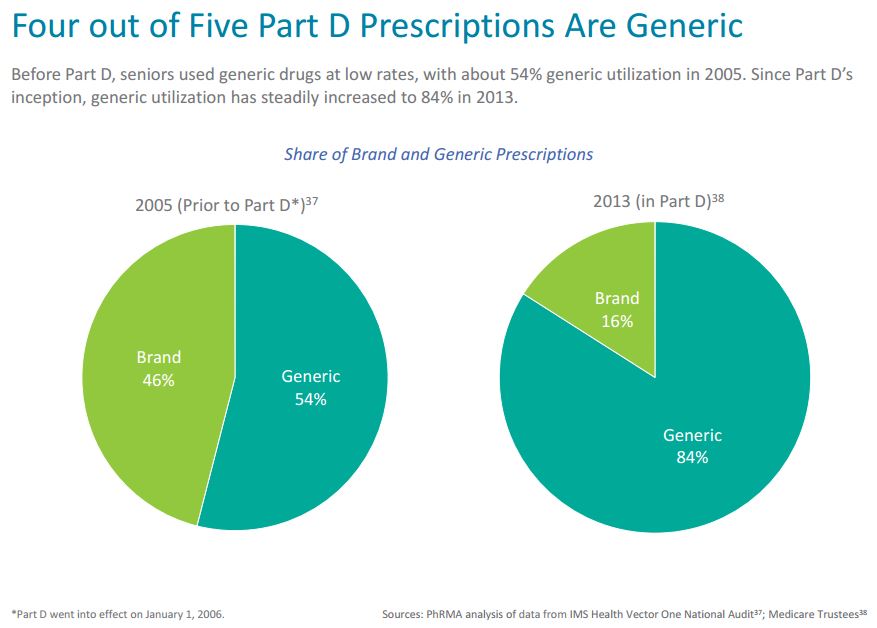
 The success of the Medicare Part D program has been well documented, providing seniors and individuals living with disabilities access to affordable prescription drug coverage. Before the implementation of Medicare Part D, seniors used generic medicines at low rates, with about 54 percent generic utilization in 2005. Since Part D’s implementation and the creation of a marketplace for older Americans to access needed medicines, generic utilization has increased steadily to 84 percent in 2013 and 85 percent in 2014 (as recently reported by the Medicare Trustees).
The success of the Medicare Part D program has been well documented, providing seniors and individuals living with disabilities access to affordable prescription drug coverage. Before the implementation of Medicare Part D, seniors used generic medicines at low rates, with about 54 percent generic utilization in 2005. Since Part D’s implementation and the creation of a marketplace for older Americans to access needed medicines, generic utilization has increased steadily to 84 percent in 2013 and 85 percent in 2014 (as recently reported by the Medicare Trustees).
Premiums for Part D plans have been stable since 2011, and last week CMS announced that average monthly premiums for 2016 Part D plans will again remain stable, maintaining affordable access to prescription drug coverage for seniors and individuals living with disabilities.
To learn more on the biopharmaceutical industry research and development lifecycle visit Innovation.org.
Read additional Medicare related posts.
Subscribe to updates [immediate, daily, weekly or monthly] here. {{cta('579cfd36-897d-4d7c-90cd-6ec935ead1f9')}}


 The
The 
