Almost all of us have fought – or know someone who is fighting – the ruthless disease author Siddhartha Mukherjee termed, “the emperor of all maladies.” Today, cancers remain the second-leading cause of death in the United States, and it is estimated that more than 1.6 million new cases of the disease will be diagnosed this year alone. That’s half the population of Alexandria, Egypt – where cancer was first discovered, around 1500 B.C.[1]
Fortunately, awareness, prevention and treatment options have dramatically improved in recent decades, spelling remarkable progress against one of mankind’s toughest medical challenges.
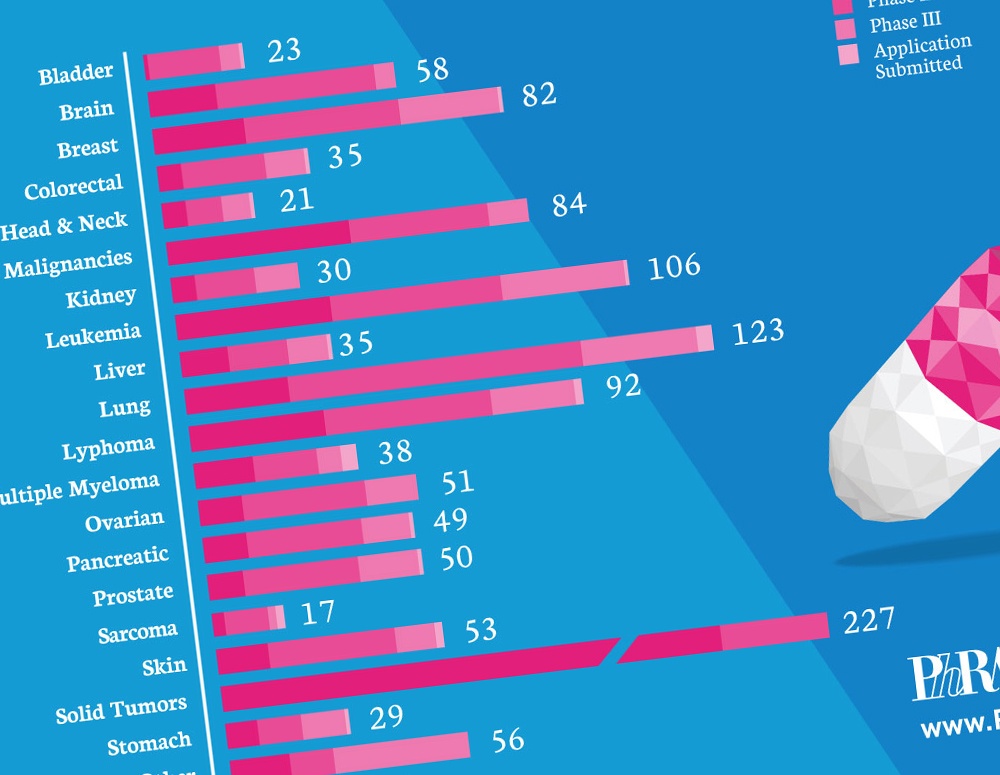
According to a new PhRMA report released today, America’s biopharmaceutical research companies are currently developing 836 medicines and vaccines to treat a broad range of cancers. This is more than have ever been in development globally.
These medicines, all of which are either in clinical trials or awaiting review by the U.S. Food and Drug Administration, include:
- 123 for lung cancer, the leading cause of cancer death in the United States, with more than 158,000 deaths expected in 2015;
- 106 for several types of leukemia, which account for 3 to 4 percent of all new cases of cancer;
- 92 for lymphoma, including non-Hodgkin lymphoma, which accounts for nearly 9 percent of all new cancer diagnoses;
- 82 for breast cancer, the leading cancer diagnosed in U.S. women, with 231,840 new cases expected in 2015;
- 58 for brain tumors, including gliomas, which represent 80 percent of all malignant tumors; and,
- 53 for skin cancer, including melanoma, which accounts for 2 percent of all skin cancer cases, but most of the skin cancer deaths.
These efforts to discover new, more effective treatments represent our best hope for helping cancer patients live longer, healthier lives while continuing to lessen the burden of this disease worldwide.
To celebrate the progress and promise of oncology research and care, PhRMA honored oncology researchers and patient advocates yesterday at the 2015 Research & Hope Awards in Washington, D.C.. Read more about the award recipients at http://www.phrma.org/research-hope-awards.
[1] http://www.cancer.org/cancer/cancerbasics/thehistoryofcancer/the-history-of-cancer-what-is-cancer
{{cta('13a5edf4-90de-4653-a862-46954152f58b','justifycenter')}}
{{cta('98bf8279-d07a-43f2-a1f6-a0d6f93af01f','justifycenter')}}