Last week, the Biden administration announced support for a waiver of the World Trade Organization (WTO) Trade-Related Aspects of Intellectual Property Rights (TRIPS) Agreement, which could lead to the removal of intellectual property protections for manufacturers of COVID-19 vaccines and treatments – a move some have suggested will speed global access in countries devastated by recent surges.
However, once you peel back the layers of the TRIPS waiver and understand the risks to health and safety, recent data shows that Americans want a better way. In fact, a recent Hill-HarrisX poll found that a majority of Americans – 57% – oppose the waiver. Recent polling conducted by Morning Consult on behalf of PhRMA echoed these strong concerns around the dangerous consequences of waiving intellectual property – including risks to public safety and vaccine manufacturing – and explored alternatives that could better expand global access to life-saving vaccines and treatments. Below are three notable findings:
1. While Americans want to expand global access to vaccines, they would prefer to build on successful U.S. manufacturing rather than waive patents.
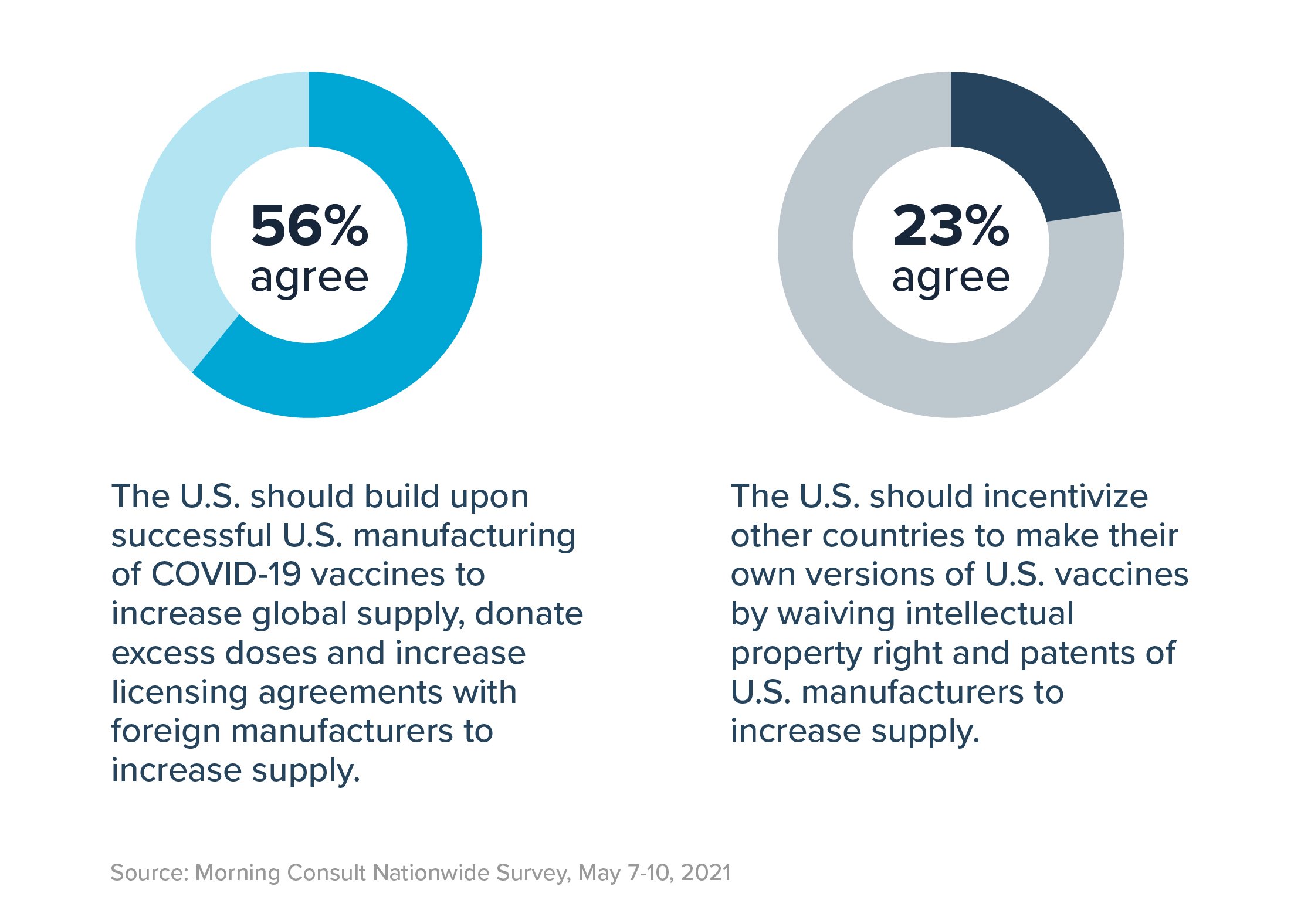
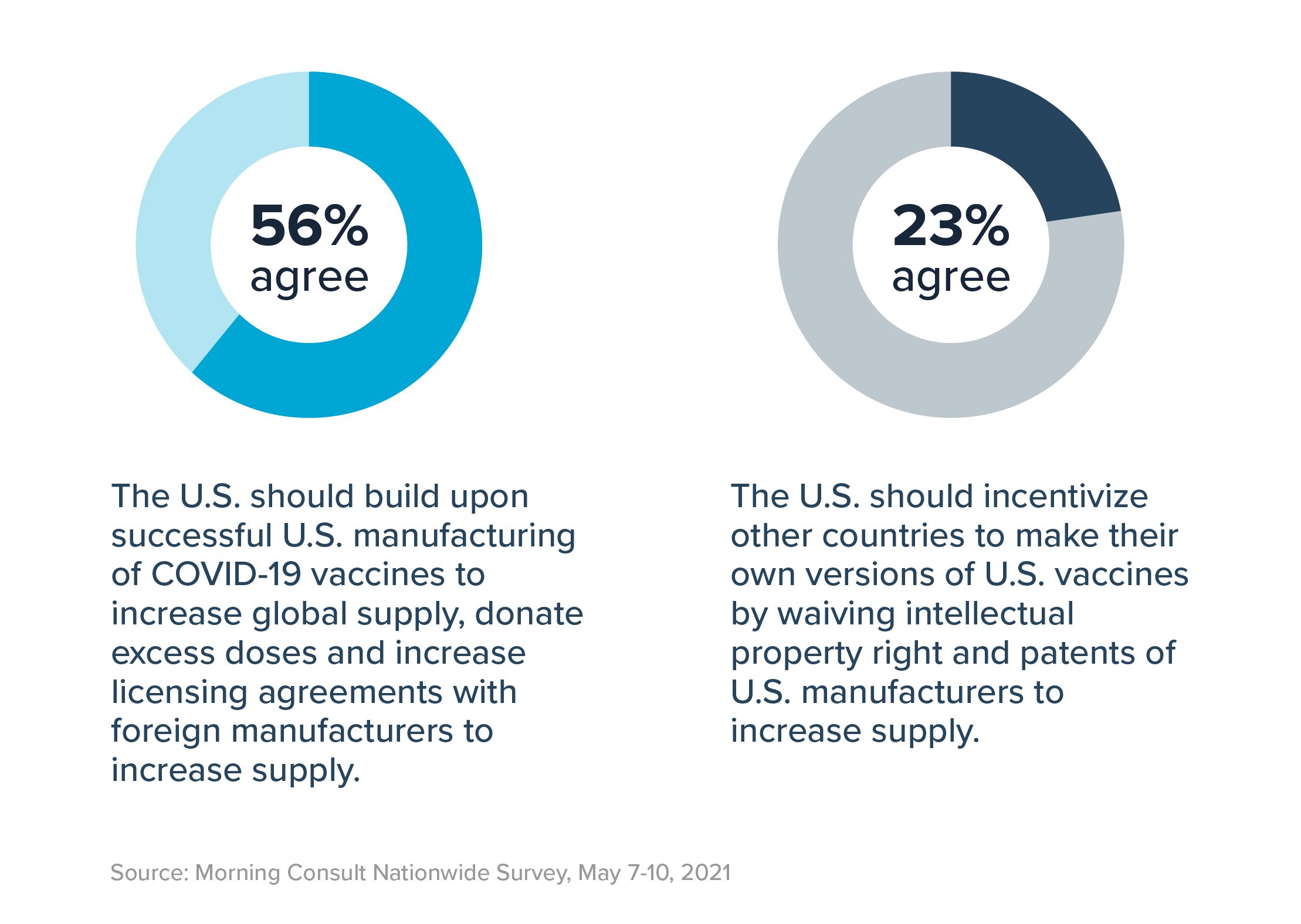
Americans support expanding vaccine access globally. In fact, 70% of registered voters believe it is a “top priority” to ensure COVID-19 vaccines become widely available for all countries, including 82% of Democrats and 56% of Republicans.
However, they would prefer to build on existing U.S. manufacturing efforts rather than undermine patents through the TRIPS waiver. When asked to choose between the two, a strong majority – 56% – prefers that we “build upon successful U.S. manufacturing of COVID-19 vaccines to increase global supply, donate excess doses and increase licensing agreements with foreign manufacturers to increase supply.” Conversely, only 23% of voters support waiving intellectual property rights of U.S. manufacturers as the means to that end.
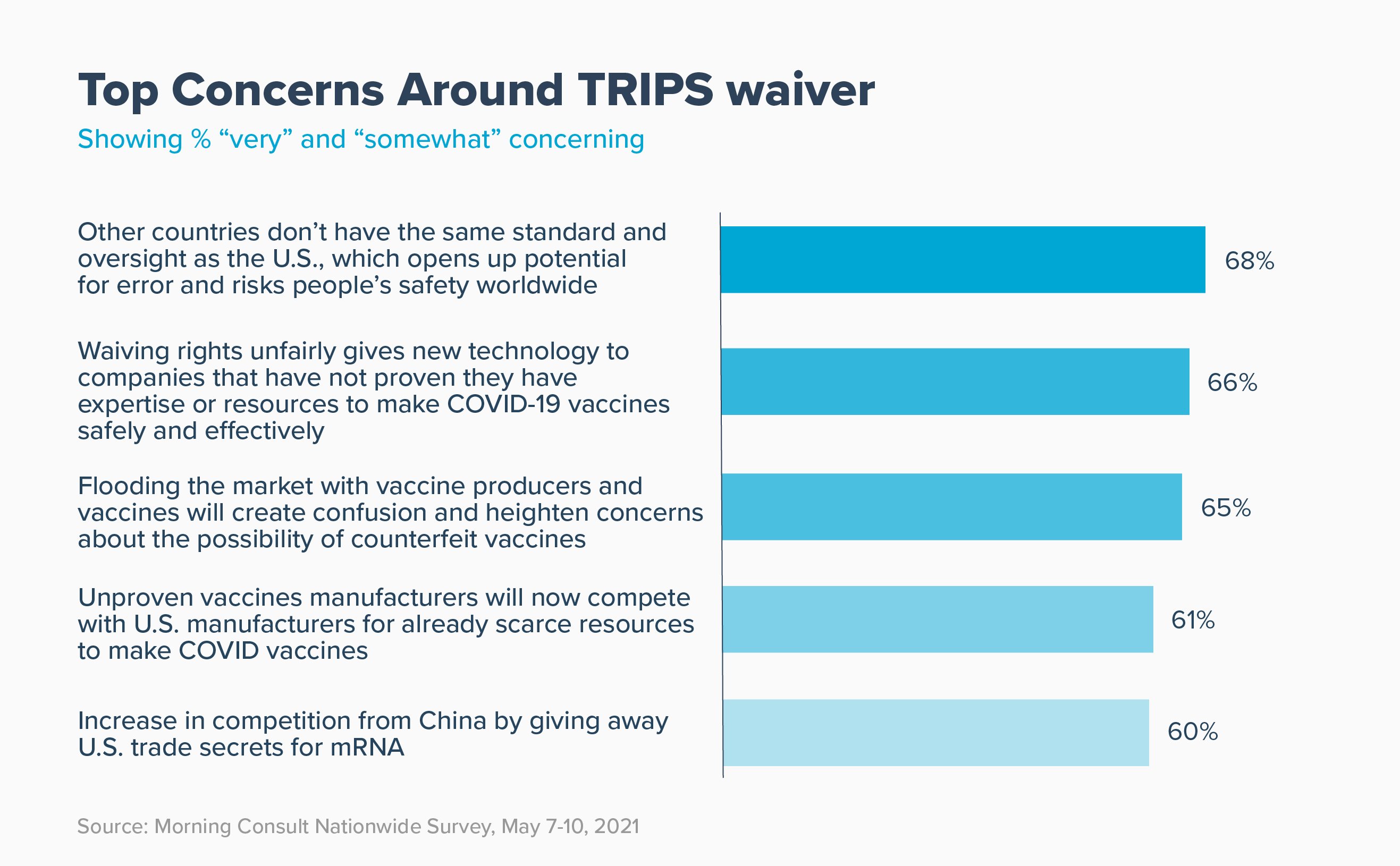
2. Americans are concerned that the TRIPS waiver could risk patient safety, sow public confusion, and cede America’s global innovation leadership to China.
Americans worry that waiving intellectual property introduces unnecessary and dangerous risks to safety and vaccine manufacturing. The top concerns – expressed by more than six in ten voters – include the following:
3. Americans want strong intellectual property and more collaboration to expand global access.
Americans across party lines support intellectual property protections for COVID-19 vaccines. In fact, three-quarters (75%) believe that the U.S. needs strong intellectual property protections to encourage innovation.
In looking to solutions, 4-in-5 voters (80%) say that vaccine manufacturers working together to help get vaccines to poorer countries is important. This collaboration was the top-ranked alternative to the TRIPS waiver for supporters of both parties: 84% support among Democrats and 74% support among Republicans. Voters also strongly support the following approaches to help increase global COVID-19 vaccine and treatment access:
- Increasing production targets from manufacturers (73%);
- U.S. vaccine developers partnering directly with manufacturers in poorer countries (71%);
- Providing financial support and in-kind donations to local organizations in poorer countries (69%);
- Increasing American vaccine exports (69%); and
- Making new commitments to the global vaccine equity initiative COVAX (68%).
Americans want a better way toward COVID-19 vaccine and treatment equity than what the TRIPS waiver would offer: one that involves protecting strong intellectual property, building on the United States’ innovation and manufacturing leadership to date, increasing collaboration, and addressing the true impediments to access, such as infrastructure, last-mile distribution and trade barriers.
Click here for detailed findings.