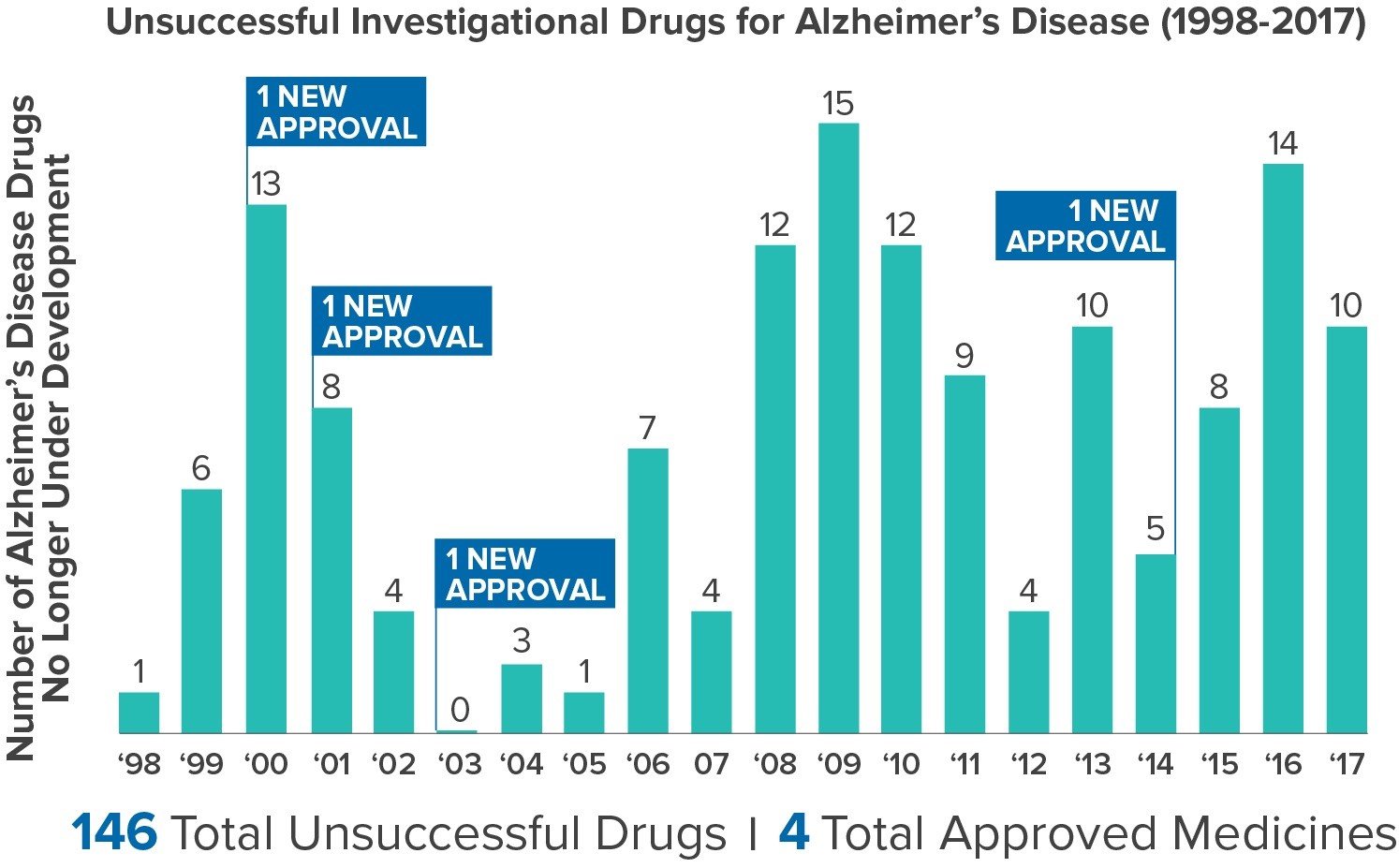
Illustrating the challenges of developing medicines to fight Alzheimer’s disease, a new analysis found that between 1998 and 2017, there were 146 unsuccessful medicines in clinical trials for Alzheimer’s. In that same time frame, only four new medicines were approved to treat the symptoms of Alzheimer’s disease. In other words, for every successful research project, about 37 failed to yield a new medicine – a 2.7 percent success rate.
In the world of Alzheimer’s research, headlines announcing failed drug candidates are all too common. Patients, caregivers, health care providers and researchers all experience the disappointment that comes with these setbacks. But it’s also important to recognize that with these so-called failures comes new information that brings us one step closer to a disease-modifying medicine.
Researchers are learning from past setbacks and are actively pursuing all promising avenues for tackling Alzheimer’s. Biopharmaceutical companies are currently researching 92 new potential medicines for the treatment of Alzheimer’s disease. And those medicines attack the disease in diverse ways that researchers hope will halt or prevent Alzheimer’s. An analysis by UsAgainstAlzheimer’s of medicines in development found that of those in Phases II and III, approximately 75 percent have the potential to be disease-modifying treatments.
That progress is sorely needed. For the 5.7 million Americans currently living with Alzheimer’s disease, their caregivers and the health care system at large, the human and economic costs are daunting. This terrible disease robs a person of everything they hold dear: their memories, their relationships, their personality, their independence and, ultimately, their life. It costs society $277 billion a year in direct medical costs, and about one in every five Medicare dollars is spent on Alzheimer’s care.
The report also details the unique challenges of researching and treating Alzheimer’s, where diagnosis is difficult, and we lack a full understanding of the disease. Successful disease-modifying treatments continue to elude the research community for several reasons, including limitations of preclinical models; the absence of validated, non-invasive biomarkers; clinical trial challenges; and, above all, a significant gap in scientific knowledge.
As the aging population expands, so does the need to address and conquer Alzheimer’s disease. Without future progress, the cost and resource burden of Alzheimer’s will continue to grow, and many more individuals and families will be impacted. However, the continued commitment of biopharmaceutical companies and other research partners, brings hope that we will meet the significant research challenges, change the course of this disease and, ultimately, prevent Alzheimer’s patients from becoming Alzheimer’s patients in the first place.
To read the full report, click here.




