Cardiovascular disease, which includes heart disease and stroke, is the leading cause of death in the United States, accounting for one out of every three deaths. According to the American Heart Association, there are more than 800,000 cardiovascular disease deaths in the United States each year and 92 million Americans are living with at least one form of cardiovascular disease or the after effects of a stroke.
Despite these staggering figures, there has been tremendous progress in recent years in the prevention, diagnosis and treatment of cardiovascular disease. Greater education and awareness about lifestyle and behavioral changes, coupled with significant treatment advances, are helping people with heart disease survive and live longer, healthier lives. And there are many exciting innovative medicines on the horizon.
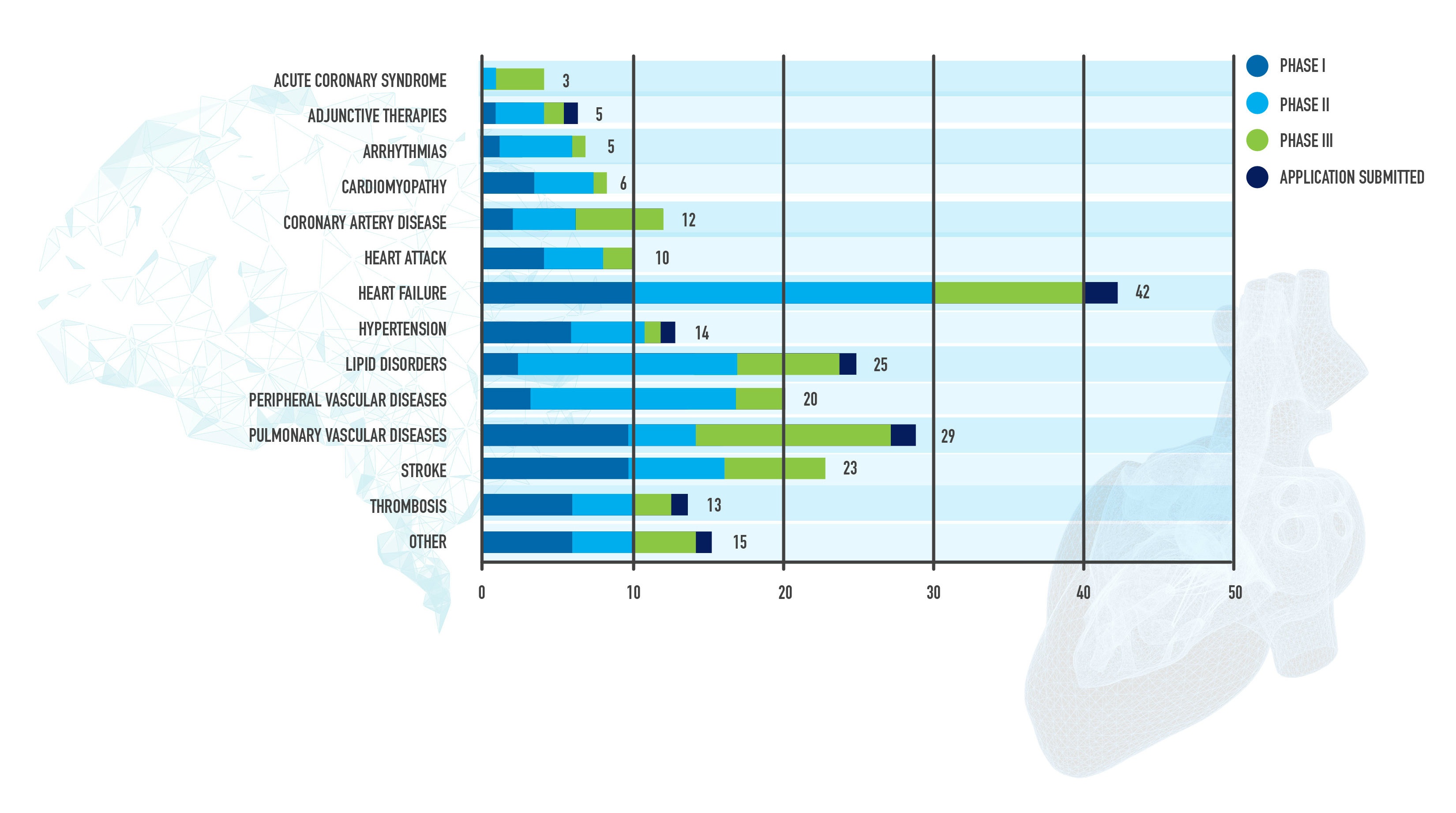
Today, a new report shows there are currently 200 medicines in development in the U.S. by America’s biopharmaceutical companies for cardiovascular diseases, including 42 for heart failure, 23 for stroke and 14 for hypertension or high blood pressure.
![MID_Diseases_Phases_Chart_FINAL[1] FOR POSTING 2.jpg](/-/media/Project/PhRMA/PhRMA-Org/HubspotImages/hubfs/464546/MID_Diseases_Phases_Chart_FINAL-1--FOR-POSTING-2.jpg)
Medicines in the biopharmaceutical pipeline include several novel approaches to treat cardiovascular disease. Among the 200 medicines in development are potentially new approaches for treating:
- Coronary Artery Disease: A medicine is in clinical trials to potentially reduce major adverse cardiac events, such as heart attack or stroke, in patients with coronary artery disease, type 2 diabetes and low levels of “good” cholesterol.
- Heart Failure: A non-viral gene therapy is in development to treat ischemic heart failure by targeting a tissue repair and regeneration pathway in the body.
- Stroke: A novel treatment is being tested with the potential to reverse brain damage suffered from a stroke.
As part of PhRMA’s GoBoldly campaign, we are sharing patient stories like Roxanne’s to help raise awareness that new treatments have significant lowered the death rates in the U.S. for heart disease and stroke. To learn more about Roxanne and the biopharmaceutical innovations helping the millions of Americans with heart disease click here.


![MID_Diseases_Phases_Chart_FINAL[1] FOR POSTING 2.jpg](/-/media/Project/PhRMA/PhRMA-Org/HubspotImages/hubfs/464546/MID_Diseases_Phases_Chart_FINAL-1--FOR-POSTING-2.jpg)
