Diabetes represents a significant public health issue in the U.S. It affects more than 30 million Americans, including around 7.2 million who are undiagnosed. Additionally, there are close to 1.5 million new cases diagnosed annually, and projections suggest the number of those impacted by the disease will continue to grow.
Diabetes treatment has improved dramatically over the years, with the availability of a range of insulin therapies more closely resembling insulin released naturally in the body as well as other antidiabetic medicines that help patients more effectively manage their condition. The options available today help meet a wide range of unmet needs, providing patients with the tools necessary to stay adherent and healthy—saving costs throughout the health care system. Yet even with these advances, diabetes remains a complex disease to successfully manage and treat.
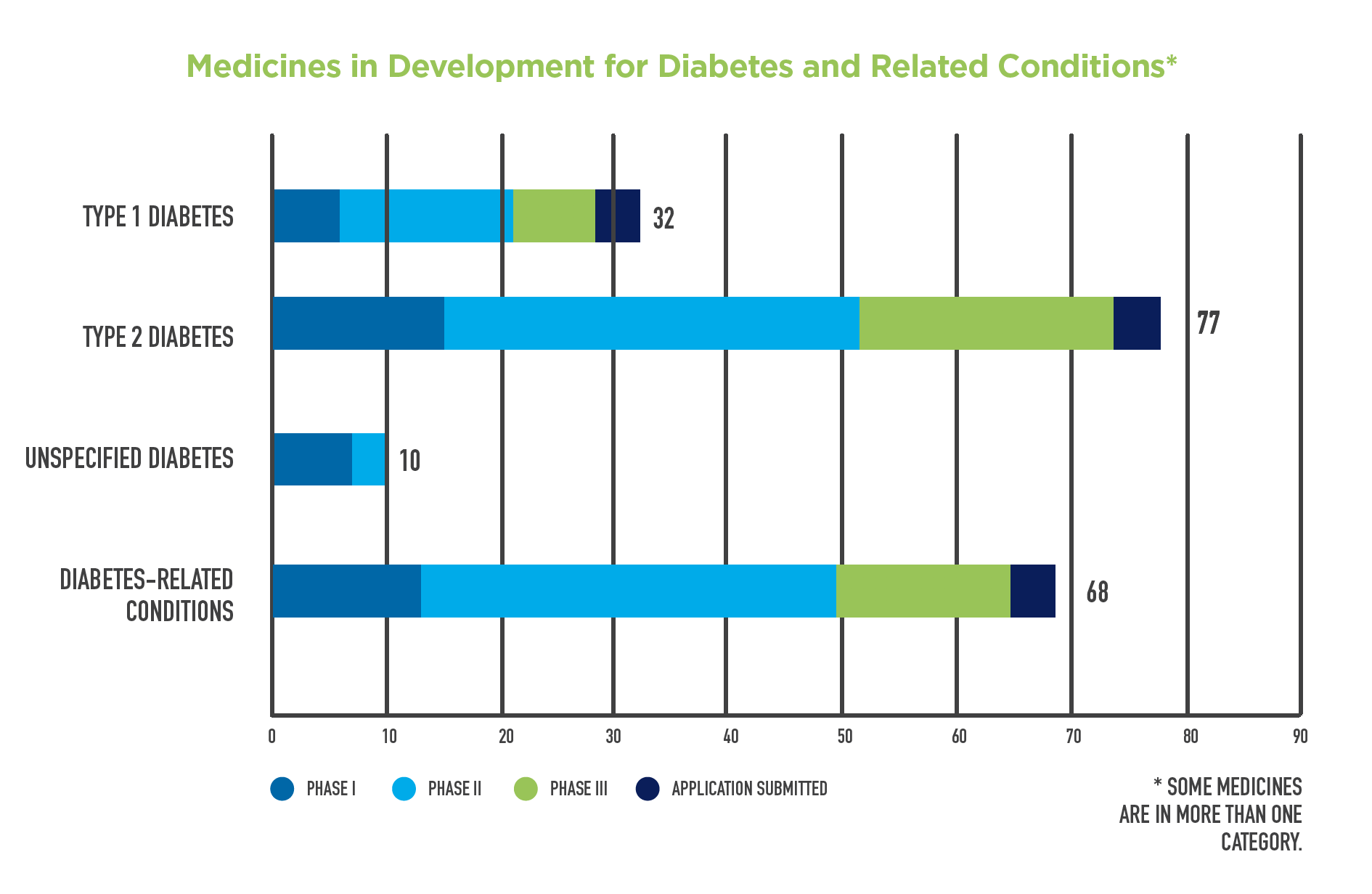
A new report released today reveals tremendous promise in the diabetes pipeline with 167 innovative new treatments in development for type 1 and type 2 diabetes and related conditions.
As PhRMA President and CEO Stephen J. Ubl highlights in the report, “Researchers and scientists are working on cutting-edge treatments that will make managing diabetes easier, and we are fighting for ways to make these innovations more affordable for patients.”
The 167 innovative medicines in development for type 1 and type 2 diabetes and related conditions include:
- 77 medicines for type 2 diabetes: In type 2 diabetes, which comprises up to 95 percent of diagnosed diabetes cases, the body is resistant to the action of insulin. To combat this resistance, the pancreas makes even more insulin until it fails to produce enough insulin to overcome the resistance, causing blood glucose levels to be higher than normal.
- 32 medicines for type 1 diabetes: Type 1 diabetes, which comprises about 5 percent of diagnosed diabetes cases, is an autoimmune disease where the body does not produce insulin as a result of the immune system attacking the insulin-producing cells of the pancreas. It is usually diagnosed in children and young adults and requires lifelong insulin treatment for survival.
- 68 medicines for diabetes-related conditions: These include chronic kidney disease due to diabetes (diabetic nephropathy), painful diabetic neuropathy, diabetic macular edema and diabetic gastroparesis.
The 167 medicines in development for diabetes and related conditions represent a heightened understanding of the unique circumstances of each patient and help pave the way for a more promising future for disease treatment.
Despite continuous advances in diabetes treatments, too many patients struggle to afford their insulin and other medicines at the pharmacy counter. Even though prices for insulin after discounts and rebates have fallen in recent years, these negotiated savings are often not shared with patients, resulting in patients with diabetes paying higher out-of-pocket costs for their insulin than their insurer. The biopharmaceutical industry is committed to improving patient access and affordability for medicines, and ensuring treatments meet the needs of all diabetes patients.
For more information on the facts of diabetes, visit PhRMA.org/Diabetes.




