
Potential personalized medicines represent 42 percent of drugs in the pipeline. This new finding, from a survey by the Tufts Center for the Study for Drug Development (CSDD), is remarkably high, particularly given that about 15 years ago the science of genomic medicine was just developing and that number was virtually zero.
Until recently many believed that biopharmaceutical companies were resistant to personalized (or precision) medicine and were not performing research to advance the field. Critics often speculated that the science was too hard and companies would not consider developing medicines for such small patient populations.

In 2010, a survey by Tufts CSDD helped to dispel this myth and, last week, the new study, sponsored by the Personalized Medicine Coalition, showed continued momentum and growth in the pipeline.
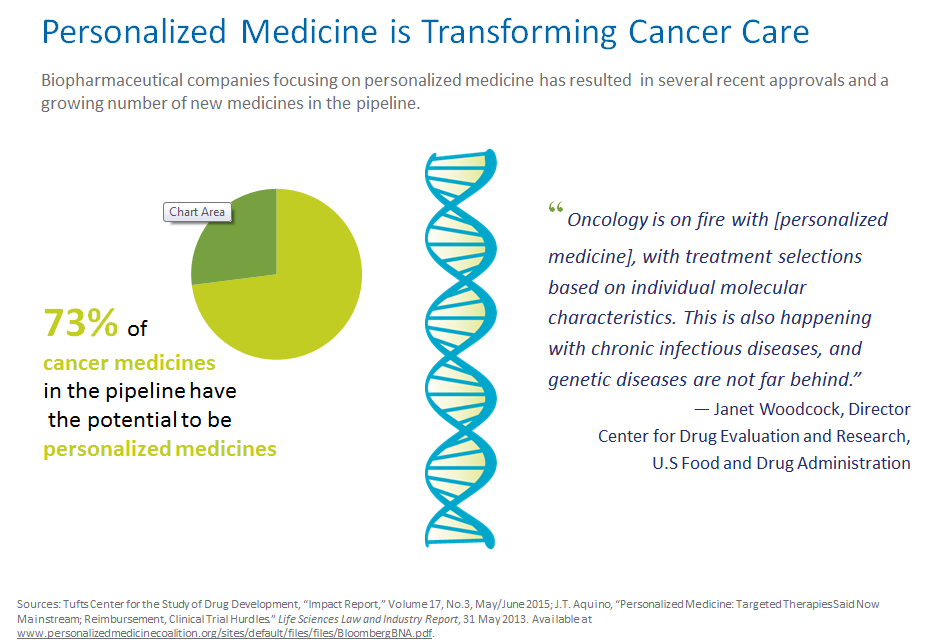
- Oncology leads the way with 73 percent of the pipeline represented by personalized therapies.
- Biopharmaceutical companies nearly doubled their R&D investment in personalized medicines over the past five years, and expect to increase their investment by an additional one-third in the next five years.
- Biopharmaceutical researchers forecast a 69 percent increase in the number of personalized medicines in development over the next five years.
Recent approvals have shown the fruits of this work. In 2014, 20 percent of medicines approved were personalized, treating diseases like ovarian cancer, lung cancer, and the rare Gaucher disease, among others. These treatments promise to improve patients’ lives – and the health care system – by getting the right medicine to each patient more quickly.
Despite this robust progress, many challenges remain. The Tufts CSDD survey and interviews revealed that companies view major challenges not only in the science, but also in the regulatory and reimbursement realms. However, with policies that foster innovation and address the unique barriers to personalized medicine, we will continue to see progress and deliver on the promising personalized medicine pipeline.
Get updates on Personalized Medicine and other issues here. {{cta('579cfd36-897d-4d7c-90cd-6ec935ead1f9')}}






