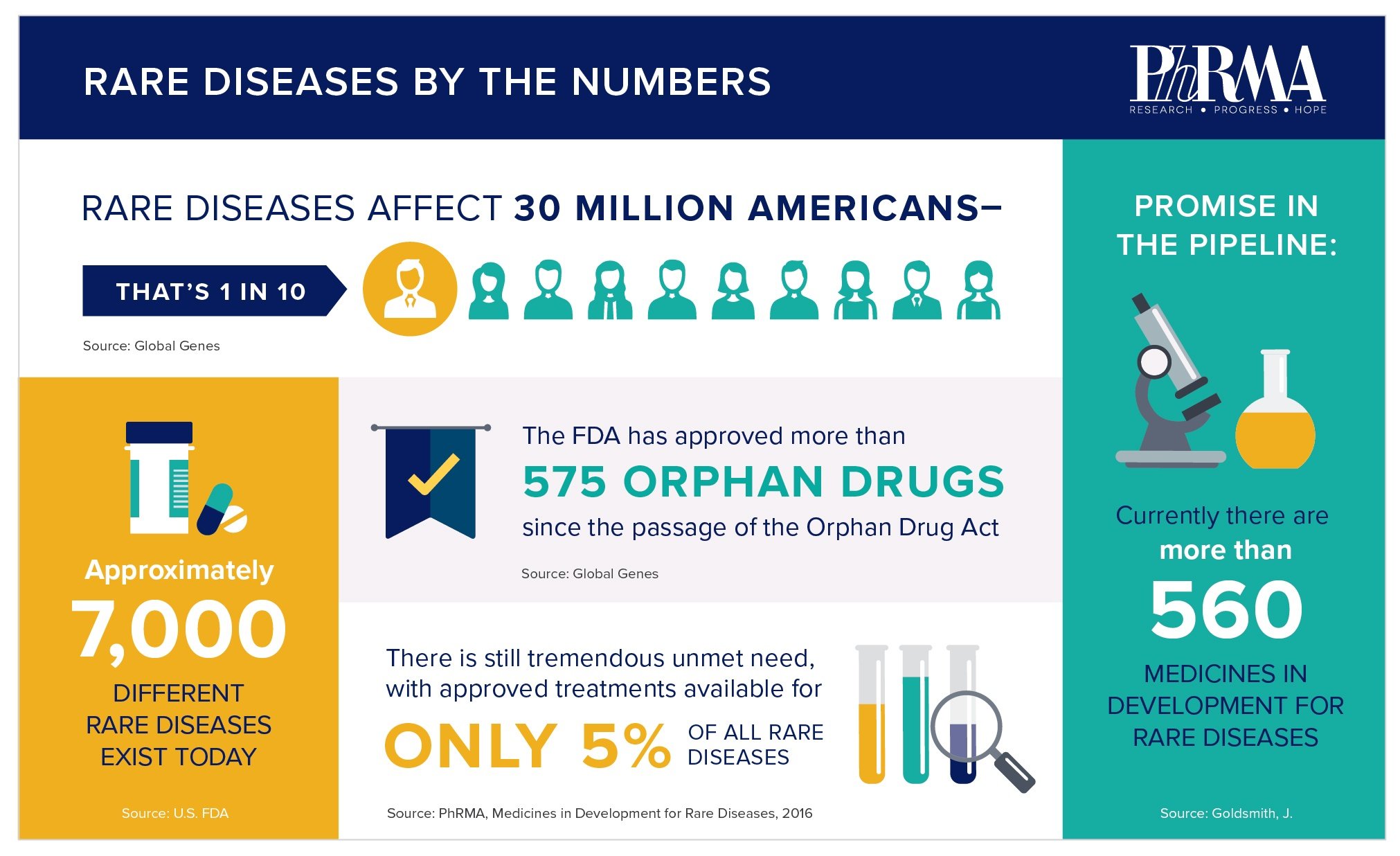
As we celebrate Rare Disease Day, it is important to not only reflect on how far we have come in tackling these devastating and complex conditions, but how much unmet medical remains. While more than 7,000 rare diseases have been identified, only 5 percent have treatments. As the majority of these diseases are life threatening, these facts underscore the need for new medicines.
 Conquering rare diseases presents a variety of unique challenges for scientists and researchers. The underlying causes of rare diseases can be difficult to identity and the symptoms of the diseases are often quite different for each patient, making rare diseases very difficult to diagnose. We now know that more than 80 percent of rare diseases are genetic in origin, giving us greater insights into the biological drivers of many conditions. Progress in understanding some of these diseases at the molecular and genomic level is helping to make new advances possible but for many rare diseases the causes remain elusive. Even within a particular rare disease, there can be many variations or subtypes resulting in different clinical manifestations and disease progression.
Conquering rare diseases presents a variety of unique challenges for scientists and researchers. The underlying causes of rare diseases can be difficult to identity and the symptoms of the diseases are often quite different for each patient, making rare diseases very difficult to diagnose. We now know that more than 80 percent of rare diseases are genetic in origin, giving us greater insights into the biological drivers of many conditions. Progress in understanding some of these diseases at the molecular and genomic level is helping to make new advances possible but for many rare diseases the causes remain elusive. Even within a particular rare disease, there can be many variations or subtypes resulting in different clinical manifestations and disease progression.
Conducting clinical research in rare diseases is also made more difficult as a result of the practicalities of recruiting and enrolling very small populations of patients. Although a rare disease is generally defined as affecting fewer than 200,000 people in the United States, research suggests that the median population size for rare diseases is actually fewer than 10,000 patients.
The Orphan Drug Act continues to be an important force in driving treatment innovation for rare diseases. More than 575 orphan drugs have been approved since its passage in 1983. In contrast, the FDA had approved fewer than 10 orphan drugs in all of the 1970s before the Act was passed. Unprecedented scientific potential, combined with appropriate incentives, make this a promising time for many patients with rare diseases.
At the same time, we also believe that enhancements to regulatory science offer to accelerate and add clarity to the research and development of treatments for rare diseases, as well as the FDA review of innovative treatments. Advancing the use of patient-focused drug development tools, novel clinical trial designs, and emerging, qualified biomarkers can help reduce uncertainty in the drug development process and ensure that researchers are incorporating the patient perspective into each aspect of the development and review of new medicines.
The biopharmaceutical industry is committed to advancing novel therapies for the more than 30 million Americans suffering from a rare disease today. Together with stakeholders across the rare disease community, biopharma scientists and other researchers are working to overcome often significant research and development challenges to drive innovative options for patients. There are more than 560 medicines currently in development for rare diseases, including more than 150 for rare forms of cancer.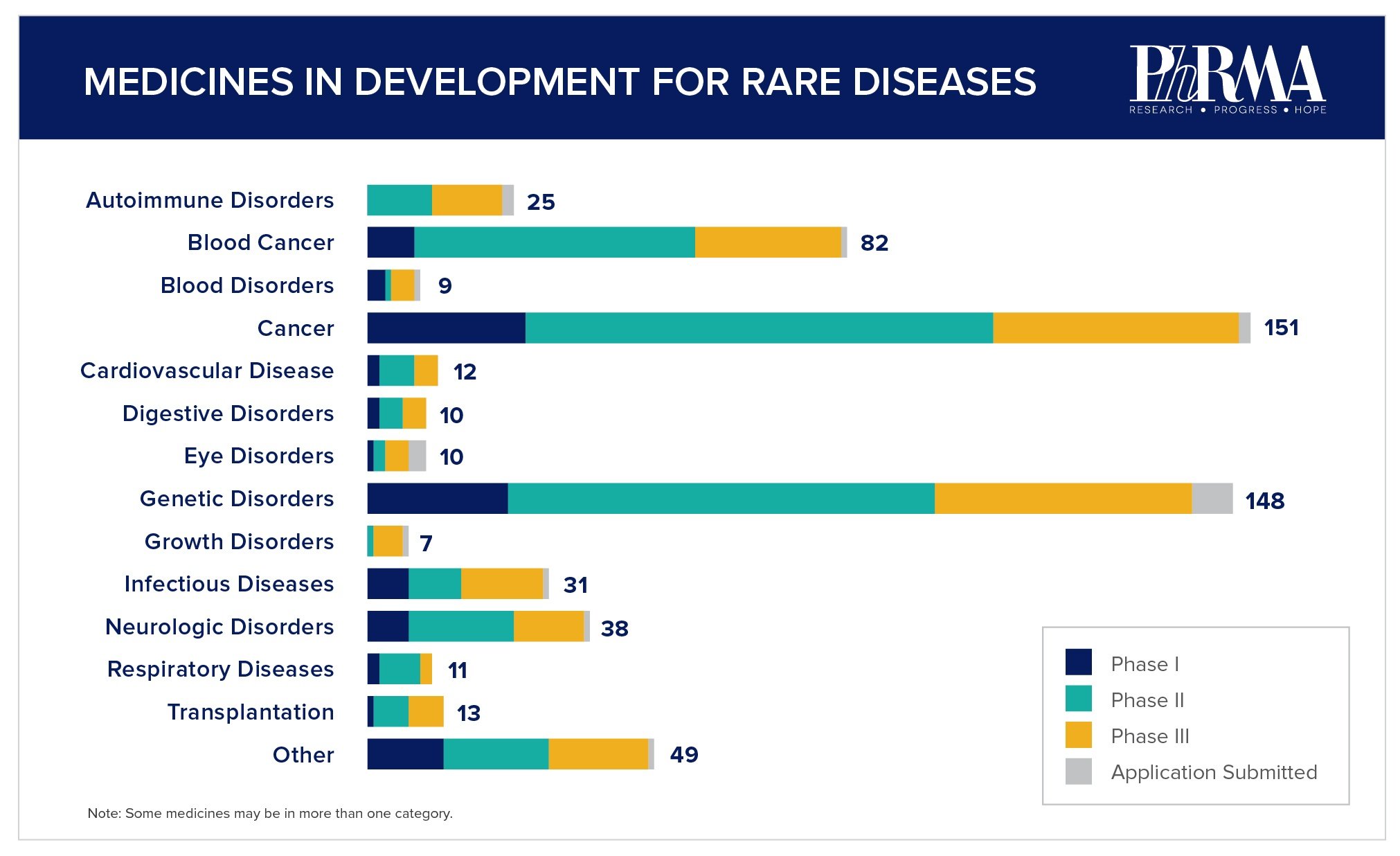
Learn more about progress in rare disease research here.
Be sure to check the Catalyst this week as we’ll be spotlighting perspectives from the rare disease community.



 Conquering rare diseases presents a variety of unique challenges for scientists and researchers. The underlying causes of rare diseases can be difficult to identity and the symptoms of the diseases are often quite different for each patient, making rare diseases very difficult to diagnose. We now know that more than 80 percent of rare diseases are genetic in origin, giving us greater insights into the biological drivers of many conditions. Progress in understanding some of these diseases at the molecular and genomic level is helping to make new advances possible but for many rare diseases the causes remain elusive. Even within a particular rare disease, there can be many variations or subtypes resulting in different clinical manifestations and disease progression.
Conquering rare diseases presents a variety of unique challenges for scientists and researchers. The underlying causes of rare diseases can be difficult to identity and the symptoms of the diseases are often quite different for each patient, making rare diseases very difficult to diagnose. We now know that more than 80 percent of rare diseases are genetic in origin, giving us greater insights into the biological drivers of many conditions. Progress in understanding some of these diseases at the molecular and genomic level is helping to make new advances possible but for many rare diseases the causes remain elusive. Even within a particular rare disease, there can be many variations or subtypes resulting in different clinical manifestations and disease progression.
