Today, we celebrate Rare Disease Day to both reflect on the progress made toward conquering rare diseases and recognize the continued unmet medical need for these devastating, complex conditions.
A rare or orphan disease is defined as affecting fewer than 200,000 people in the United States. However, with more than 7,000 rare diseases identified to date, when examined together, rare disease patients are anything but rare. According to the National Institutes of Health (NIH), 30 million Americans, or 10 percent of the population, have a rare disease.
Patients with rare disease have seen significant advances in recent years. The Orphan Drug Act (ODA) has been instrumental in driving innovation for the treatment of rare diseases and increasing rare disease research. Since ODA passed in 1983, more than 600 orphan drugs and biological products have been approved in the United States. In recent years, groundbreaking therapies have included:
- The first treatments directed at treating the underlying causes of cystic fibrosis.
- New medicines that can prevent or slow the impact of extremely rare, devastating conditions including pulmonary arterial hypertension, hereditary angioedema and Gaucher disease.
- The first therapies available to treat rare pediatric diseases, including a progressive, metabolic disease called hypophosphatasia (HPP), an inherited genetic disease called lysosomal acid lipase (LAL) deficiency, and neuroblastoma, a rare form of cancer that occurs in nerve cells and the brain.
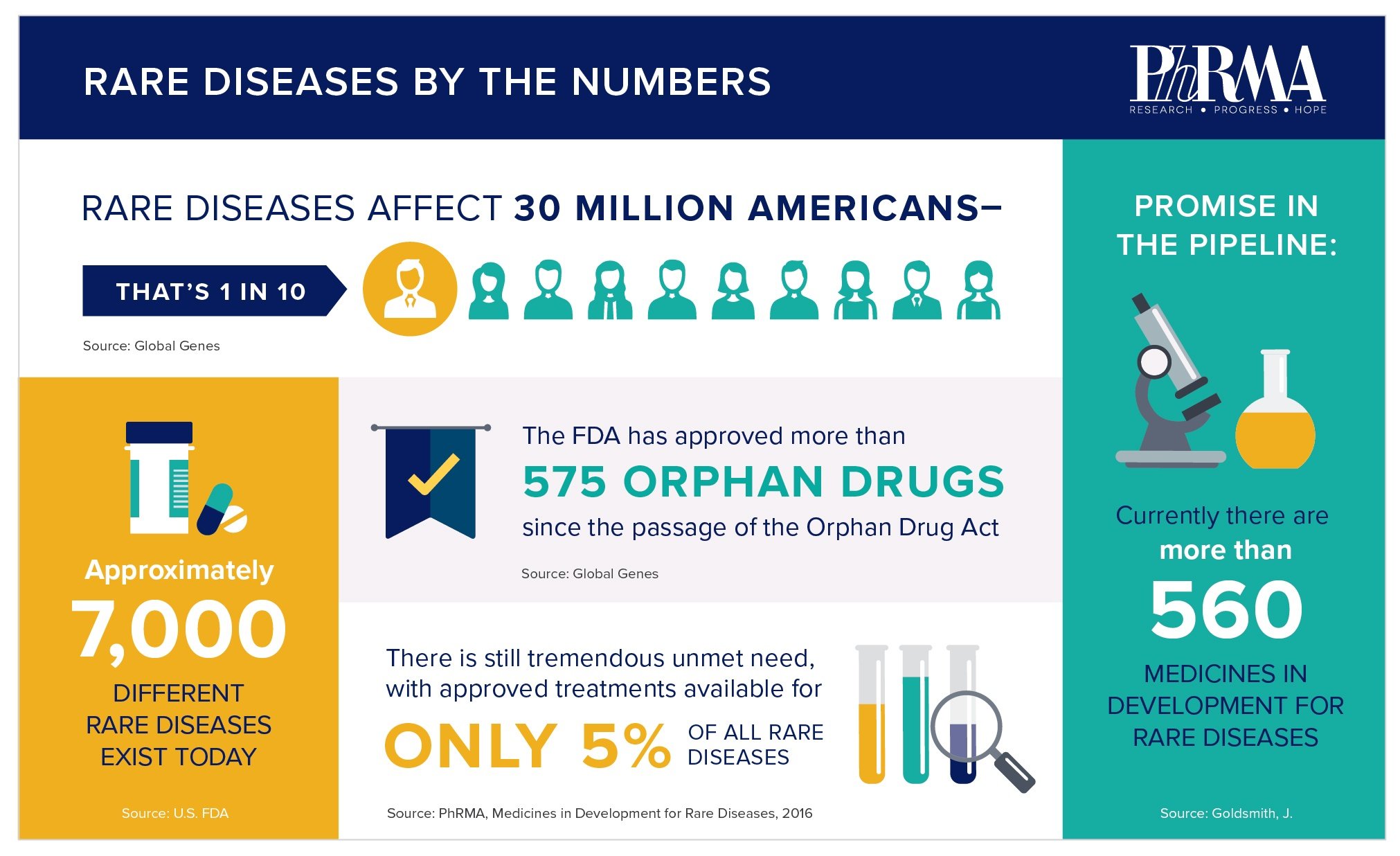
While these advances have been very impactful, there remains tremendous unmet need, as only five percent of rare diseases have an approved treatment option. Developing treatments for these diseases is a challenge due to the complex biology of the diseases and inherently small patient populations, making it difficult for researchers to design and implement a drug development program and conduct clinical trials.
The biopharmaceutical industry is committed to overcoming these challenges and advancing new medicines for the millions of patients with rare diseases. The biopharmaceutical pipeline contains thousands of significant and innovative new treatments with the potential to address rare diseases. In fact, the recent report, “The Biopharmaceutical Pipeline: Innovative Therapies in Clinical Development,” found that 822 projects are designated by the FDA as orphan drugs, and are currently in clinical development or regulatory review.




