You are probably reading a lot about a surge of cases of respiratory viruses like respiratory syncytial virus (RSV), influenza (the flu) and SARS-CoV-2, which causes COVID-19. The triple threat of these highly contagious respiratory viruses circulating simultaneously is causing a ripple of effects on patients, health care providers and our health care system as a whole.
We’ve discussed on this blog before that thankfully, for both the flu and COVID-19, we have effective vaccines and therapeutics that are approved or authorized for prevention and treatment of both diseases. Just this past weekend, I was excited to take my 8- and 10-year-old kids for their annual flu shot and their bivalent COVID-19 booster right in time for the cold season. Unfortunately, with respect to RSV, there are fewer existing tools in the toolbox.
As an emergency physician, some of the scariest moments I’ve had were caring for infants experiencing respiratory distress caused by RSV. And as a parent, one of the scariest moments for me was in the winter of 2014, when I took care of my own son while he was sick with RSV at only eight months old. He was teetering on the edge of having serious respiratory problems, but thankfully recovered quickly. For too many families, they aren’t as lucky.
But there is hope. In the same way that I caught chickenpox when I was a kid while my younger brother didn’t (thanks to the chickenpox vaccine), my hope for parents in the future is that they won’t need to worry about RSV in the same way that we are worrying about it now. And that is possible.
Thanks to the commitment of the biopharmaceutical industry, key investments have been made over decades to better understand and target RSV. While the cellular structure of the virus makes it a challenging nemesis, there are a number of potential medicines on the horizon. Currently there is one U.S. Food and Drug Administration (FDA)-approved monoclonal antibody that can be prescribed to high-risk infants and children to help reduce the risk of RSV-related hospitalizations. And excitingly, there are 21 medicines and vaccines currently in development.
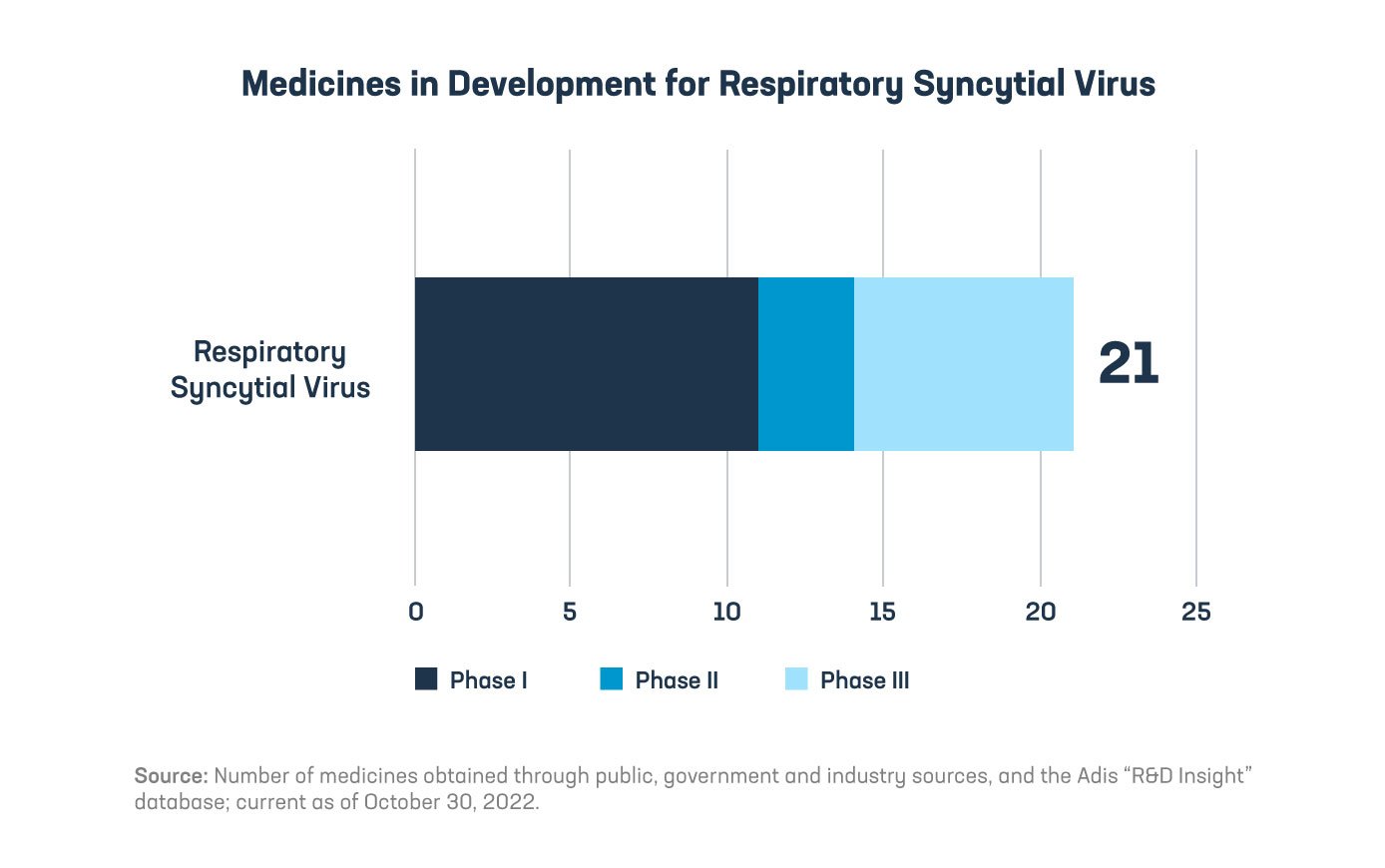
Several RSV vaccines in the pipeline are for three at risk populations: pregnant women (who can pass along immune protection to their babies in utero), infants and older adults. These potential vaccines are groundbreaking and diverse in terms of their mechanisms of action, and some have received FDA breakthrough designations for regulatory review. Some of the vaccine candidates are even building on our knowledge from COVID-19 mRNA vaccines. Antiviral treatments are also in development to reduce the severity of illness for individuals that catch RSV.
All of this innovation is possible because of investments the biopharmaceutical industry has made and partnerships they have developed across the life sciences ecosystem. This is not by accident, but because we have chosen to take risks and seek answers to the most vexing scientific and medical challenges. At the same time, policymakers should prioritize policies like strong intellectual property (IP) protections which support, not hinder, drug development. Unfortunately, the Inflation Reduction Act’s Medicare price-setting provisions gut the incentives necessary to encourage risky biopharmaceutical research and development, essentially devaluing IP by allowing for government price controls to take effect years before the product’s patents or regulatory exclusivities expire.
While we anxiously await new innovations to change the paradigm for RSV, there are actions you can take today to keep you and your family safe and healthy. Consider getting up to date on influenza and COVID-19 vaccinations. Continue to do the simple things also like washing your hands, wearing a mask in crowded indoor spaces and staying home when you feel sick. Mild cold symptoms in a healthy adult could be a serious and life-threatening RSV infection for an infant.
Everyone doing their part will go a long way to controlling the spread of these contagious respiratory illnesses and keeping at-risk individuals healthy this holiday season.




