
If a home inspector found “wide and unpredictable variation” in your smoke detectors, would you want to fix them?
For any policymaker serious about the issues of access and affordability, a new analysis issued by Avalere should raise the same issue regarding the budget impact threshold used by the Institute for Clinical and Economic Review (ICER). While this issue sounds arcane, it holds real implications for stakeholders, particularly patients. If ICER gets its thresholds wrong, it could have serious, negative implications for patient access to important new treatments.
Avalere found that “wide and arbitrary variation in ICER’s budget threshold may limit its reliability as a benchmark.” In particular, Avalere found that the budget impact threshold calculated by ICER ($904 million from 2015 to 2016) can change dramatically based on the year a new medicine is approved by the Food and Drug Administration (FDA). That’s because one of the variables ICER uses to calculate the budget impact threshold is annual FDA drug approvals, which varies considerably from year to year.
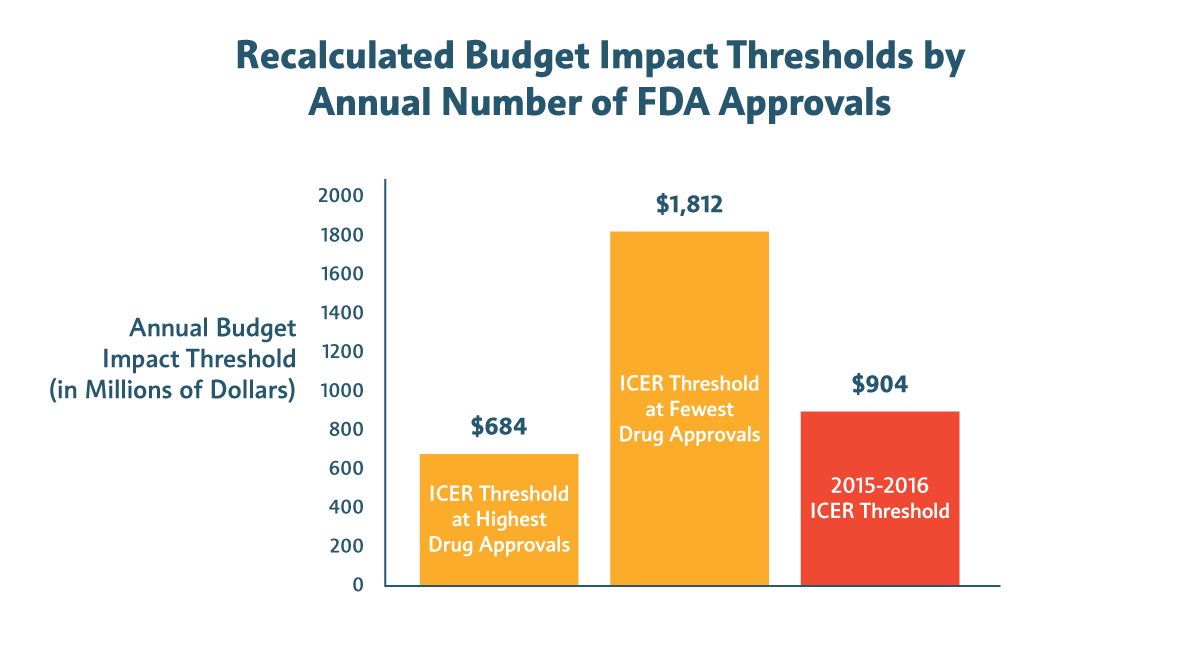
For example:
- In years with fewer FDA approvals, 94 percent to 100 percent of patients with non-small cell lung cancer could be treated with new medicines without triggering a budget impact affordability alert. But during years of high approvals, fewer than 40 percent of patients in need could receive these drugs before crossing ICER’s threshold.
- Slightly less than half of patients with plaque psoriasis would have access to new treatments in the best-case scenario. In years of high approvals, the percentage of patients who could be treated before triggering an affordability alert would be just 26 percent and 29 percent, respectively.
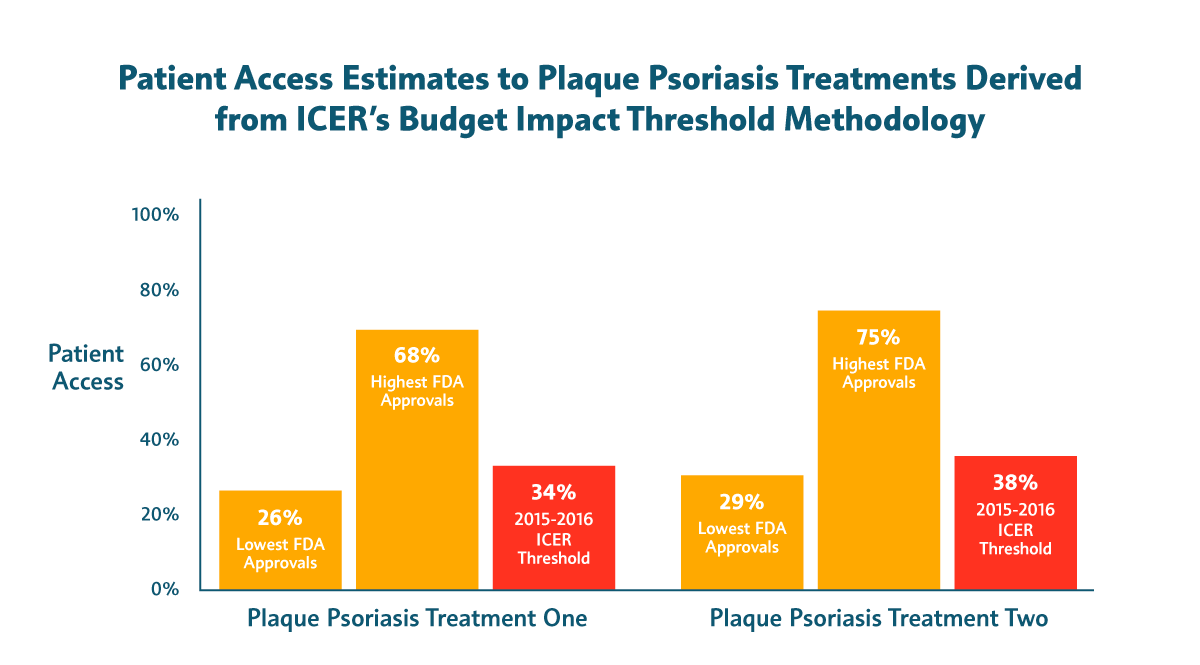
Patients deserve better than access restrictions based on false alarms. That’s why our industry frequently provides payers data on the estimated budget impact of new treatments and why our sector is making unprecedented investments in better tools for value assessment.
View the Avalere study here.






