In the not too distant past, chemotherapy and radiation were once the only treatments for what remains the second leading cause of death in the United States – cancer. Although the death rates of cancer have declined in the past 20 years and survival rates have increased by five times since the 1970s, there is still more work to be done. The biopharmaceutical industry is leading by translating science into a completely new era of medicine for how we treat the diseases which make up cancer.
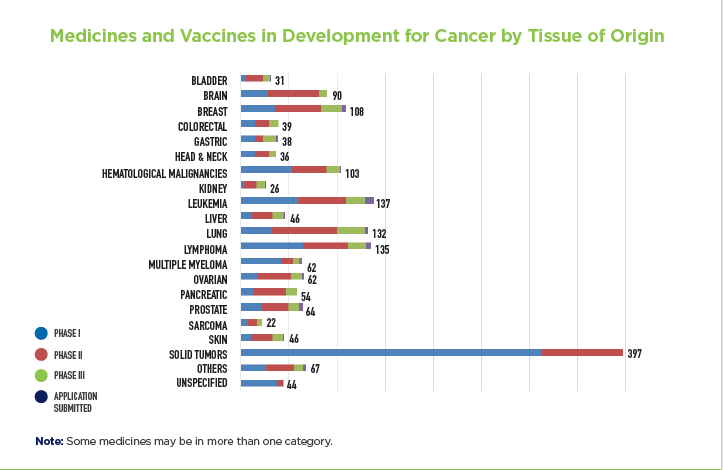
In a new Medicines in Development report released today, as a result of unprecedented advancements in science, there are currently 1,120 new medicines and vaccines in development to treat cancer – all of which are in clinical trials or awaiting review by the U.S. Food and Drug Administration (FDA).
Immuno-oncology and personalized medicines are ushering in a new era of cancer understanding and treatment. Immunotherapies like CAR-T and checkpoint inhibitors are further harnessing the body’s own immune system to recognize and kill cancerous tumor cells. Even in just the last year, 47 new immuno-oncology treatments have been added to the development pipeline.
Innovation is critical for improving patients’ lives, and at the same time we need innovation to deliver these medicines to patients. No patient should struggle to afford their needed treatments, and it is important that we address patient access challenges. On the societal level cancer treatments are only about one percent of total health care spending and looking ahead, total drug spending growth is moderate, which includes cancer spending.
Our new chart pack puts cancer costs in context for patients and society, and puts forward solutions for improving on our current system.
The 1,120 medicines and vaccines in development represent an increased recognition among researchers that no two cancers are alike, which has led to the creation of treatments to target specific cancer types, and cancers specific to a single person. These medicines represent treatments across the cancer spectrum, from lung cancer to leukemia, breast cancer to lymphoma. For a full list of these medicines click here.






