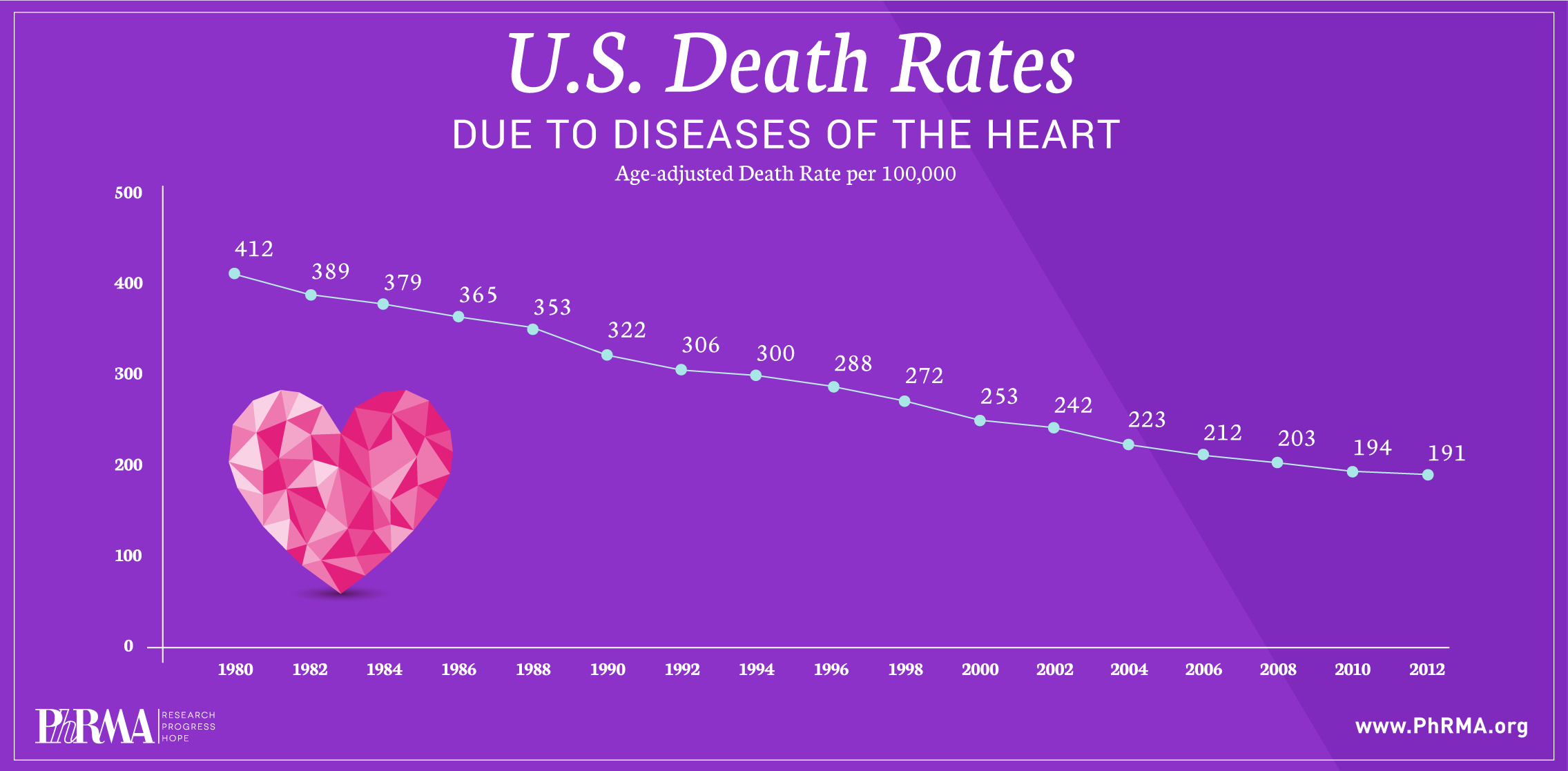
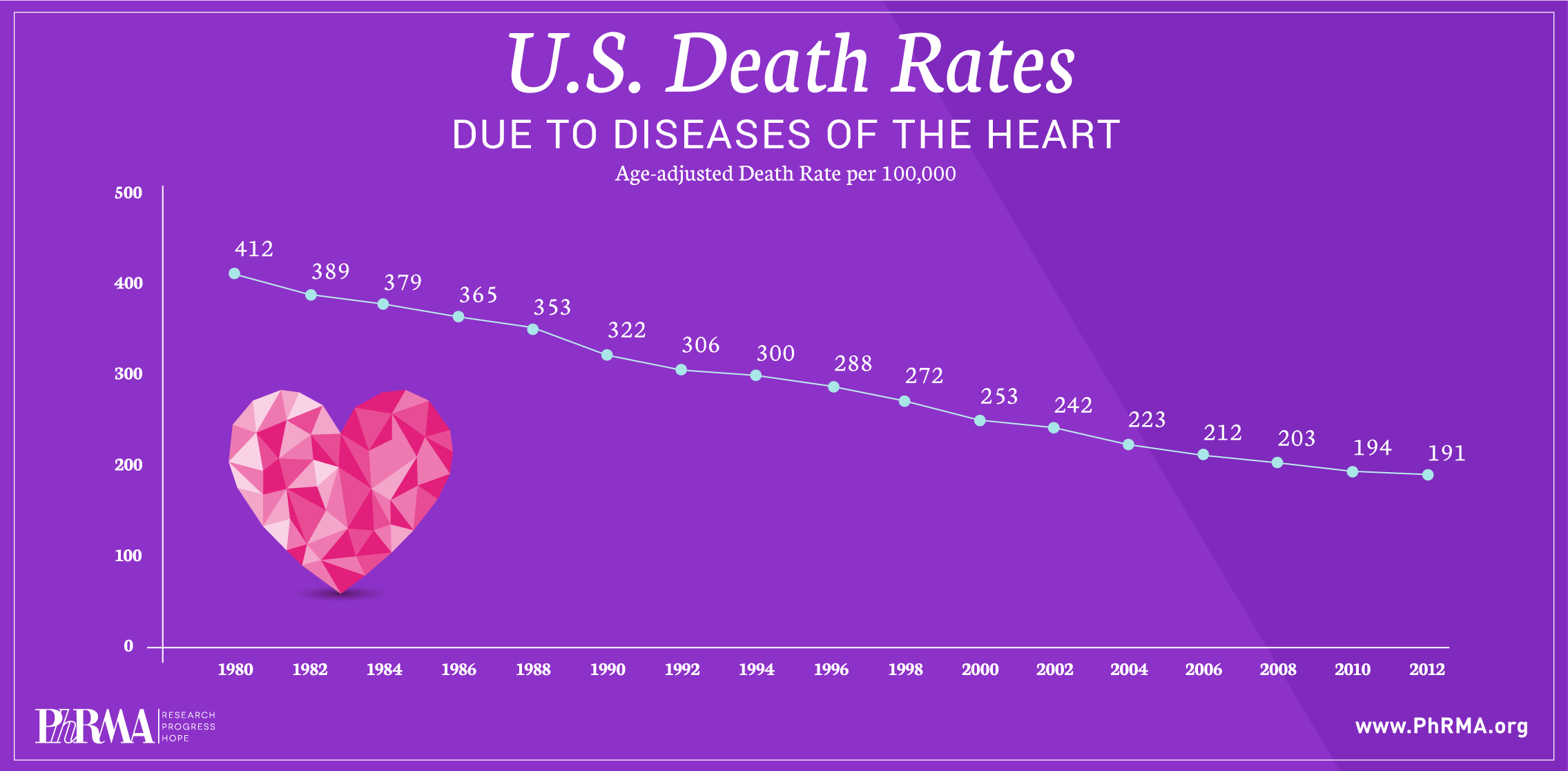
Despite significant decreases in death rates from heart disease and stroke over the past three decades, these two conditions continue to be leading causes of death in the United States.

With more than 85 million Americans living with some type of cardiovascular disease, including more than six million adults who have had a stroke, chances are that you or someone you know would stand to benefit from new medicines that help prevent and treat these illnesses.
Fortunately, a new report released today from the Pharmaceutical Research and Manufacturers of America (PhRMA) and the Association of Black Cardiologists (ABC) shows that America’s biopharmaceutical companies are currently developing 190 medicines for heart disease and stroke – many of which will build on progress made by existing treatments.
These medicines, all of which are either in clinical trials or awaiting review by the U.S. Food and Drug Administration (FDA), include:
- 33 for heart failure, which affects approximately 5.7 million American adults;
- 29 for lipid disorders, including high cholesterol, which affects 30.9 million American adults with total cholesterol levels of more than 240;
- 21 for stroke, which affects 6.6 million American adults;
- 18 for peripheral vascular diseases, including critical limb ischemia and intermittent claudication;
- 12 for hypertension, or high blood pressure, which affects 80 million American adults; and,
- 12 for thrombosis, a condition characterized by coagulation or clotting of the blood in the veins or arteries (circulatory system).
A critical component of progressing research and development for heart disease and stroke medicines is clinical trial enrollment. To help drive that process, ABC established a Clinical Trials Network (CTN) that matches investigators with trials and helps recruit and retain African American patients for participation.
The combined efforts of innovative companies developing new medicines, along with the support of groups like ABC, creates hope that we can bring new treatments to people in need even faster while continuing to improve patient outcomes and survival rates.
View the full report here: Medicines in Development for Cardiovascular Diseases
View the medicine list here: Heart Disease, Stroke Drug List