Currently, more than 23.5 million Americans are affected by one of the more than 80 autoimmune diseases identified to date. These diseases range in severity, with their own characteristics and their own struggles. Because of this, the need for innovative treatments and cures has never been greater. But today, patients with an autoimmune disease can have more hope than ever before with the tremendous progress that science offers patients and their families.
A new PhRMA report, “Medicines in Development for Autoimmune Diseases,” released in partnership with the Lupus Foundation of America, demonstrates how the more than 300 medicines currently in development offer patients with diseases such as arthritis, Crohn’s disease, lupus, type 1 diabetes and multiple sclerosis the opportunity to live longer, healthier lives through innovation.
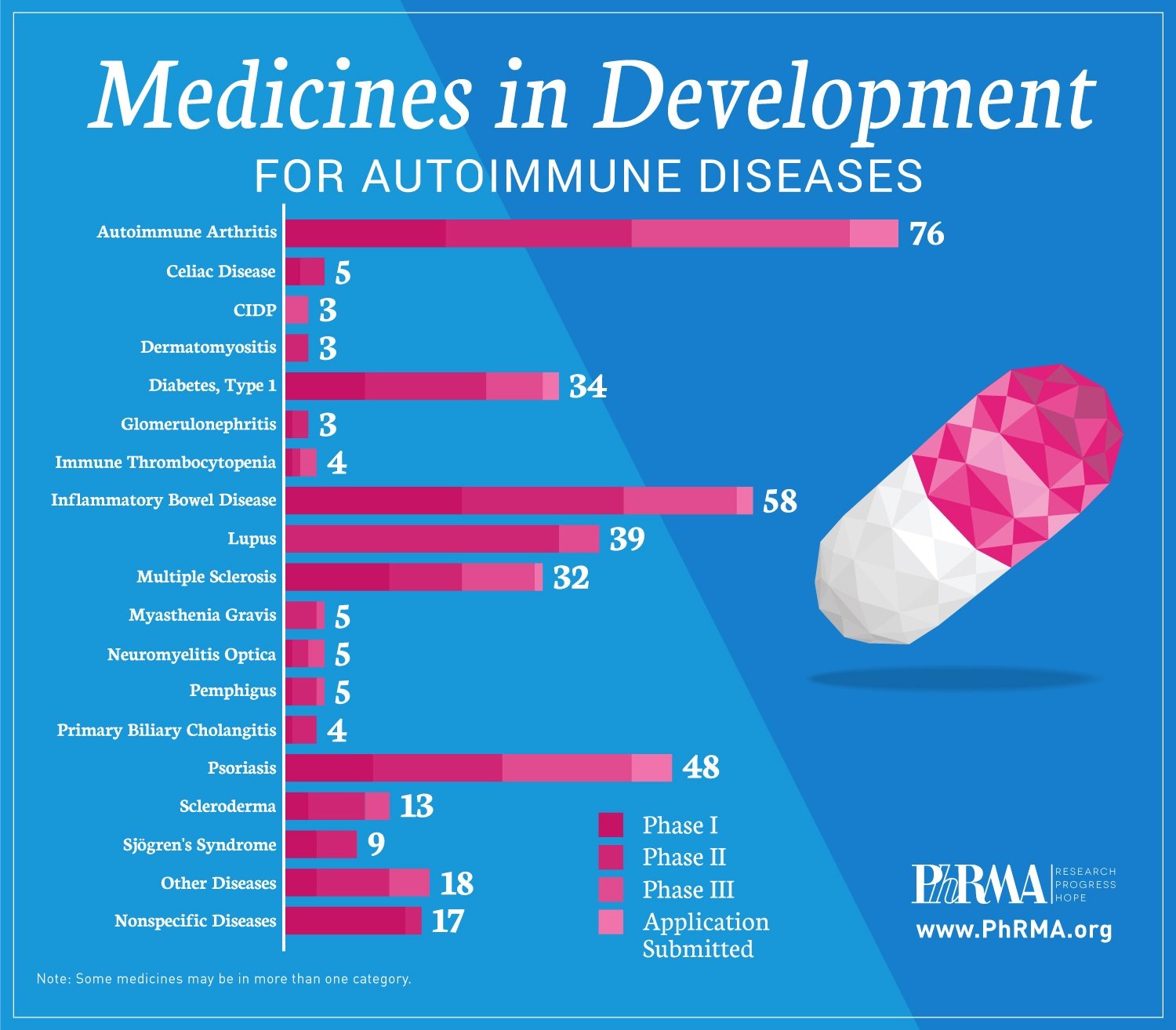
These medicines in development include:
- 76 for autoimmune types of arthritis;
- 58 for inflammatory bowel disease;
- 39 for lupus;
- 34 for type 1 diabetes; and
- 32 for multiple sclerosis.
Lupus, which affects an estimated 1.5 million Americans, is just one of the numerous autoimmune diseases with few treatment options. However, new therapies for diseases like lupus move science forward and take steps toward helping patients and their families manage diseases that have a significant effect on their lives.
America’s biopharmaceutical companies are continuing to develop treatments and cures for patients living with autoimmune diseases. And in collaborating with disease groups like the Lupus Foundation of America, as well as patients across the country, all those affected by autoimmune diseases can have hope for a healthier future and a longer life.
View the full report here.




